Mean Kärger Model Water Exchange Rate in Brain
- PMID: 40534623
- PMCID: PMC12176371
- DOI: 10.1162/imag_a_00335
Mean Kärger Model Water Exchange Rate in Brain
Abstract
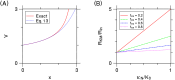
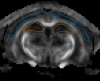
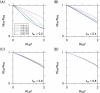
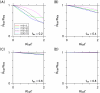
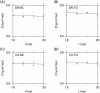
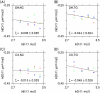
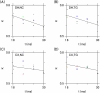
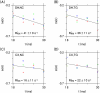
Intercellular water exchange in brain is analyzed in terms of the multi-compartment Kärger model (KM), and the mean KM water exchange rate is used as a summary statistic for characterizing the exchange processes. Prior work is extended by deriving a stronger lower bound for mean exchange rate that can be determined from the time dependence of the diffusional kurtosis. In addition, an analytic formula giving the time dependence of the kurtosis for a model of thin cylindrical neurites is demonstrated, and this formula is applied to numerically test the accuracy of the lower bound for a range of model parameters. Finally, the lower bound is measured in vivo with diffusional kurtosis imaging for the dorsal hippocampus and cerebral cortex of eight-month-old mice. From the stronger lower bound, the mean KM exchange rate is found to be or greater in dorsal hippocampus and or greater in cortex.
Keywords: Kärger model; brain; diffusion; kurtosis; mouse; water exchange.
Conflict of interest statement
DECLARATION OF COMPETING INTEREST The authors have no competing financial interests to declare.
Figures
Similar articles
-
Correcting pulse duration effects in the diffusional kurtosis of the multi-compartment Kärger model.Magn Reson Med. 2025 Nov;94(5):2249-2257. doi: 10.1002/mrm.30608. Epub 2025 Jun 10. Magn Reson Med. 2025. PMID: 40493748 Free PMC article.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
In vivo cortical microstructure mapping using high-gradient diffusion MRI accounting for intercompartmental water exchange effects.Neuroimage. 2025 Jul 1;314:121258. doi: 10.1016/j.neuroimage.2025.121258. Epub 2025 May 9. Neuroimage. 2025. PMID: 40349743 Free PMC article.
-
Immunogenicity and seroefficacy of pneumococcal conjugate vaccines: a systematic review and network meta-analysis.Health Technol Assess. 2024 Jul;28(34):1-109. doi: 10.3310/YWHA3079. Health Technol Assess. 2024. PMID: 39046101 Free PMC article.
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
Cited by
-
Linear rotationally invariant kurtosis measures from double diffusion encoding MRI.Magn Reson Imaging. 2025 Jul;120:110399. doi: 10.1016/j.mri.2025.110399. Epub 2025 Apr 26. Magn Reson Imaging. 2025. PMID: 40294767
-
Correcting pulse duration effects in the diffusional kurtosis of the multi-compartment Kärger model.Magn Reson Med. 2025 Nov;94(5):2249-2257. doi: 10.1002/mrm.30608. Epub 2025 Jun 10. Magn Reson Med. 2025. PMID: 40493748 Free PMC article.
References
-
- Abramowitz , M. , & Stegun , I. A. (Eds.). ( 1972. ). Handbook of mathematical functions: With formulas, graphs, and mathematical tables (p. 14 ). Dover Publications; . https://store.doverpublications.com/products/9780486612720?_pos=1&_sid=0...
-
- Benavides-Piccione , R. , Regalado-Reyes , M. , Fernaud-Espinosa , I. , Kastanauskaite , A. , Tapia-González , S. , León-Espinosa , G. , Rojo , C. , Insausti , R. , Segev , I. , & DeFelipe , J. ( 2020. ). Differential structure of hippocampal CA1 pyramidal neurons in the human and mouse . Cerebral Cortex , 30 ( 2 ), 730 – 752 . 10.1093/cercor/bhz122 - DOI - PubMed
-
- Boyd , S. , & Vandenberghe , L. ( 2004. ). Convex optimization (pp. 69 – 70 ). Cambridge University Press; . 10.1017/CBO9780511804441 - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Molecular Biology Databases
