Structured light imaging mesoscopy: detection of embedded morphological changes in superficial tissues
- PMID: 40534904
- PMCID: PMC12175002
- DOI: 10.1117/1.JBO.30.6.065001
Structured light imaging mesoscopy: detection of embedded morphological changes in superficial tissues
Abstract
Significance: Current paradigms for the optical characterization of layered tissues involve explicit consideration of an inverse problem which is often ill-posed and whose resolution may retain significant uncertainty. Here, we present an alternative approach, structured light imaging mesoscopy (SLIM), that leverages the inherent sensitivity of raw spatial frequency domain (SFD) reflectance measurements for the detection of embedded subsurface scattering changes in tissue.
Aim: We identify wavelength-spatial frequency ( ) combinations that provide optimal sensitivity of SFD reflectance changes originating from scattering changes in an embedded tissue layer. We specifically consider the effects of scattering changes in the superficial dermis which is a key locus of pathology for diverse skin conditions such as cancer, aging, and scleroderma.
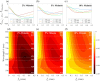
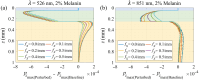
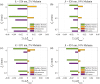
Approach: We used Monte Carlo simulations in a four-layer skin model to analyze the SFD reflectance changes resulting from changes in superficial dermal scattering across wavelength ( to 851 nm) and spatial frequency ( to 0.5/mm). Within this model, we consider different values for epidermal melanin concentration to simulate variations in skin tone.
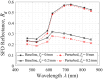
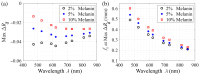
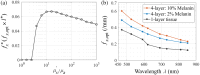
Results: Monte Carlo simulations revealed that scattering changes within the superficial dermis produce SFD reflectance changes which are maximized at specific ( ) pairs and vary with skin tone. For light skin tones, SFD reflectance changes due to scattering reductions in the superficial dermis are maximized at and spatial frequency . By contrast, for darker skin tones, maximal SFD reflectance changes occur at wavelengths in the near-infrared ( ) at a spatial frequency of . Interestingly, the change in SFD reflectance produced by such scattering changes is most uniform across all skin tones when using the longest wavelength tested ( ) and a spatial frequency of . Taken together, our computational model identifies specific ( ) pairs to optimally detect embedded structural alterations in the superficial dermis.
Conclusions: The findings establish the SLIM methodology as a means to detect morphological changes in an embedded subsurface tissue layer by leveraging inherent sensitivities of spatial frequency domain reflectance. This approach promises to enable simplified clinical tracking of subsurface microstructural alterations without the explicit need to consider an inverse problem approach.
Keywords: Monte Carlo simulation; inverse problems; perturbation methods; spatial frequency domain measurement; subsurface morphological change detection.
© 2025 The Authors.
Figures


Similar articles
-
Exploring the bias: how skin color influences oxygen saturation readings via Monte Carlo simulations.J Biomed Opt. 2024 Jun;29(Suppl 3):S33308. doi: 10.1117/1.JBO.29.S3.S33308. Epub 2024 Aug 29. J Biomed Opt. 2024. PMID: 39211937 Free PMC article.
-
Short-Term Memory Impairment.2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Jun 8. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31424720 Free Books & Documents.
-
Systemic Inflammatory Response Syndrome.2025 Jun 20. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2025 Jun 20. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 31613449 Free Books & Documents.
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
The effect of sample site and collection procedure on identification of SARS-CoV-2 infection.Cochrane Database Syst Rev. 2024 Dec 16;12(12):CD014780. doi: 10.1002/14651858.CD014780. Cochrane Database Syst Rev. 2024. PMID: 39679851 Free PMC article.
References
-
- Mehendale A. M., et al. , “Structured light imaging mesoscopy: application to skin changes in scleroderma,” Biophotonics Discov. 2(2), 025002 (2025). 10.1117/1.BIOS.2.2.025002 - DOI
-
- Yamada Y., “Light-tissue interaction and optical imaging in biomedicine,” Annu. Rev. Heat Transf. 6, 1–59 (1995). 10.1615/AnnualRevHeatTransfer.v6.30 - DOI

