Severe alpha-1 antitrypsin deficiency is associated with a higher risk of complications after first decompensation than other aetiologies of cirrhosis
- PMID: 40535554
- PMCID: PMC12174978
- DOI: 10.1016/j.jhepr.2025.101398
Severe alpha-1 antitrypsin deficiency is associated with a higher risk of complications after first decompensation than other aetiologies of cirrhosis
Abstract
Background & aims: Alpha-1 antitrypsin deficiency (AATD) causes/predisposes to advanced chronic liver disease. However, the role of the SERPINA1 Pi∗ZZ genotype in patients with decompensated cirrhosis is unclear. Thus, we evaluated the impact of the Pi∗ZZ genotype on the disease course after the first hepatic decompensation event.
Methods: We retrospectively included 59 adults with decompensated cirrhosis and severe AATD (Pi∗ZZ) from 12 European tertiary care centres. First decompensation was considered as baseline. To compare the course of AATD to other cirrhosis aetiologies, we applied propensity score matching for Child-Turcotte-Pugh (CTP) score as well as age/sex. Patients were followed until further decompensation, liver transplantation or liver-related death.
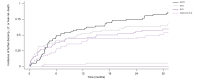
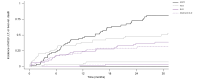
Results: Most patients were male (74.6%), with a mean age of 55 years. The most common type of first decompensation was ascites (n = 40; 67.8%), followed by variceal bleeding (n = 13; 22.0%) and overt hepatic encephalopathy (n = 6; 10.2%). Median CTP and MELD (model for end-stage liver disease) scores at first decompensation were 8 and 14, respectively. Median MELD scores were 16 and 20 points at listing and liver transplantation (median time on list: 2.9 [IQR 1.1-7.2] months), respectively. Patients with other aetiologies (subdistribution hazard ratio: steatotic liver disease: 0.62, 95% CI 0.44-0.88, p = 0.007; abstinent alcohol-associated liver disease: 0.50, 95% CI 0.35-0.71, p <0.001; hepatitis C virus-associated cirrhosis: 0.56, 95% CI 0.37-0.83, p = 0.004) had a significantly lower risk of further hepatic decompensation, liver transplantation, or liver-related death, compared to those with Pi∗ZZ. Exchanging further decompensation with acute-on-chronic liver failure yielded similar results.
Conclusion: Our study defines the course of decompensated cirrhosis in patients with severe AATD (Pi∗ZZ), who are particularly prone to complications of cirrhosis and exhibit a more progressive disease course than those with cirrhosis of other aetiologies.
Impact and implications: Alpha-1 antitrypsin deficiency is an inherited disease that affects the lung and the liver. Carrying two severely dysfunctional copies of the alpha-1 antitrypsin gene may cause advanced chronic liver disease/cirrhosis. Affected individuals with a first complication of cirrhosis are more prone to developing further liver-related events (including multiorgan dysfunction) and requiring liver transplantation (which cures the inherited liver disease) compared to patients who have similarly advanced liver disease. These findings should prompt the development of disease-modifying treatments and early listing for liver transplantation.
Keywords: AATD; acute-on-chronic liver failure; advanced chronic liver disease; ascites; hepatic encephalopathy; liver transplantation; portal hypertension; variceal bleeding.
© 2025 The Authors.
Conflict of interest statement
The authors have nothing to disclose regarding the work under consideration for publication. The following authors disclose conflicts of interests outside the submitted work: L.B. received speaker fees from Chiesi and Gilead. M.F. received consulting fees from Takeda and honoraria from CSL Behring, Grifols, and Takeda. N.K. has nothing to disclose. B.S. has nothing to disclose. S.F. received consulting fees from MSD and AOP Orphan; honoraria from AOP Orphan Gilead, and AbbVie; travel support from AbbVie and AOP Orphan; and advisory board fees from AOP Orphan. L.v.M. has nothing to disclose. J.St. has nothing to disclose. M.J. served as a speaker and/or consultant for Gilead. G.S. has nothing to disclose. B.S.H. has nothing to disclose. T.T. served as speaker for Dr. Falk Pharma. H.R. has nothing to disclose. A.S.K. has nothing to disclose. J.Sp. received consulting fees from AOP Orphan and MSD; honoraria from AOP Orphan, AbbVie and Gilead; travel support from AbbVie and AOP Orphan; and advisory board fees from AOP Orphan. M.W. has nothing to disclose. M.P. received speaker fees from CSL Behring and consulting fees from Takeda Pharamaceuticals. H.H. has nothing to disclose. M.P.-R. is advisor/consultant for AstraZeneca, Bayer, BMS, Eisai, Ipsen, Lilly, MSD, and Roche; he served as a speaker for Bayer, Eli Lilly, Eisai, Ipsen, and Roche. M.T. received speaker fees from BMS, Falk Foundation, Gilead, Intercept, Ipsen, Jannsen, Madrigal, MSD, and Roche; he advised for AbbVie, Agomab, Albireo, BiomX, Boehringer Ingelheim, Chemomab, Cymabay, Dr. Falk Pharma, Genfit, Gilead, GSK, Hightide, Intercept, Ipsen, Janssen, MSD, Novartis, Phenex, Pliant, Rectify, Regulus, Siemens, and Shire. He further received travel support from AbbVie, Dr. Falk Pharma, Gilead, Intercept, and Jannsen and research grants from Albireo, Alnylam, Cymabay, Dr. Falk Pharma, Genentech, Gilead, Intercept, MSD, Takeda, and UltraGenyx. He is also a co-inventor of patents on the medical use of norUDCA filed by the Medical Universities of Graz and Vienna. T.R. received grant support from AbbVie, Boehringer Ingelheim, Gilead, Intercept/Advanz Pharma, MSD, Myr, Philips Healthcare, Pliant, Siemens, and W. L. Gore & Associates; speaking/writing honoraria from AbbVie, Echosens, Gilead, GSK, Intercept/Advanz Pharma, MSD, Pfizer, Roche, Siemens, and W. L. Gore & Associates; consulting/advisory board fees from AbbVie, AstraZeneca, Bayer, Boehringer Ingelheim, Gilead, Intercept/Advanz Pharma, MSD, Resolution Therapeutics, and Siemens; and travel support from AbbVie, Boehringer Ingelheim, Dr. Falk Pharma, Gilead, and Roche. B.M. served as a speaker and/or advisory board member for AbbVie, Fujirebio, Gilead, Luvos, MSD, Norgine, Roche, W. L. Gore & Associates and received research support from Altona, ewimed, Fujirebio, and Roche. H.Z. received grant support from Pharmacosmos and Vifor as well as honoraria for lecturing from Pharmacosmos, Pierre Fabre, Medice, and Falk Foundation as well as for consulting from Novo Nordisk. B.v.H. has nothing to disclose. J.V. received grant support from Gilead, speaker fees from Gilead and Orphalan, travel support from AbbVie, Astellas, Dr. Falk Pharma, Gilead, Ipsen, and Janssen, as well as consultancy fees from Astellas, AstraZeneca, Eisai, Ipsen, and Takeda. P.S. received grants from Arrowhead, CSL Behring, and Grifols; consulting fees from Arrowhead, BioMarin, BridgeBio, Dicerna, GSK, Intellia, Novo Nordisk, Ono, and Takeda; honoraria from Alnylam, CSL Behring, and Grifols; and material transfer support for Vertex and Dicerna. M.M. received grant support from Echosens, served as a speaker and/or consultant and/or advisory board member for AbbVie, AstraZeneca, Echosens, Eli Lilly, Falk Foundation, Gilead, Ipsen, Takeda, and W. L. Gore & Associates, and received travel support from AbbVie and Gilead. Please refer to the accompanying ICMJE disclosure forms for further details.
Figures

References
-
- Sharp H.L., Bridges R.A., Krivit W., et al. Cirrhosis associated with alpha-1-antitrypsin deficiency: a previously unrecognized inherited disorder. J Lab Clin Med. 1969;73(6):934–939. - PubMed
-
- Eriksson S., Carlson J., Velez R. Risk of cirrhosis and primary liver cancer in alpha 1-antitrypsin deficiency. N Engl J Med. 1986;314(12):736–739. - PubMed
-
- Lomas D.A., Hurst J.R., Gooptu B. Update on alpha-1 antitrypsin deficiency: new therapies. J Hepatol. 2016;65(2):413–424. - PubMed
-
- Zoller H., Wagner S., Tilg H. Is heterozygosity for the alpha-1 antitrypsin risk allele Pi∗MZ a disease modifier or genetic risk factor? Gastroenterology. 2020;159(2):433–434. - PubMed
-
- Greene C.M., Marciniak S.J., Teckman J., et al. α1-Antitrypsin deficiency. Nat Rev Dis Primers. 2016;2 - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

