Defining Clinically Meaningful Subgroups in Patients Undergoing Arthroscopic Rotator Cuff Repair Using Unsupervised Machine Learning
- PMID: 40535587
- PMCID: PMC12174775
- DOI: 10.1177/23259671251335977
Defining Clinically Meaningful Subgroups in Patients Undergoing Arthroscopic Rotator Cuff Repair Using Unsupervised Machine Learning
Abstract
Background: Outcomes after arthroscopic rotator cuff repair (RCR) are frequently measured through clinically significant outcomes (CSOs) such as the minimal clinically important difference, the substantial clinical benefit, and the Patient Acceptable Symptom State. Global achievement of CSOs is challenging to predict.
Purpose: To determine if unsupervised machine learning can identify distinct patient subgroups based on CSO achievement after elective arthroscopic RCR.
Study design: Case-control study; Level of evidence, 3.
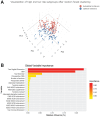
Methods: A prospectively collected database was analyzed to identify patients who underwent elective arthroscopic RCR from 2015 to 2017. Tear dimensions were measured on magnetic resonance imaging utilizing a validated technique. CSO achievements on the American Shoulder and Elbow Surgeons, the Single Assessment Numeric Evaluation, and the Constant-Murley subjective score at 2-year follow-up were calculated. An unsupervised random forest algorithm was utilized to develop and internally validate patient subgroups with significantly different rates of CSO achievement. Patient subgroup membership, along with a total of 30 demographic and clinical variables, as well as preoperative patient-reported outcomes, were incorporated into a stepwise multivariable logistic regression to identify factors predictive of optimal CSO achievement.
Results: A total of 346 patients (192 male; mean ± SD age, 57.2 ± 9.1 years; body mass index, 30.1 ± 5.4 kg/m2) were eligible for inclusion and followed for a mean of 3.8 years (range, 2.0-6.2 years) Of these, a total of 333 patients were partitioned by the random forest algorithm into 2 subgroups (stability, 0.16; connectivity: 180.8; Dunn: 0.16; silhouette: 0.05), with 176 patients in the optimal achievement subgroup and 157 patients in the suboptimal achievement subgroup. The 2 subgroups differed significantly (all P≤ .004) in the likelihood of achievement of all CSOs. Stepwise multivariable logistic regression identified an increase of 1 mm in tear size in the sagittal dimension beyond 1.9 cm to predict a 10% increase in the probability of suboptimal achievement. Additional risk factors for suboptimal CSO achievement included increasing number of tendons involved (odds ratio [OR], 14.07; 95% CI, 4.50-44.02; P < .001), subscapularis involvement (OR, 8.67; 95% CI, 2.45-30.71; P = .01), and increased preoperative CMS score (OR, 1.11; 95% CI, 1.04-1.18; P = .001). Protective factors included performance of a subpectoral biceps tenodesis compared with biceps tenotomy (OR, 0.22; 95% CI, 0.05-0.92; P = .03).
Conclusion: Clinically meaningful subgroups were uncovered using an unsupervised machine learning algorithm in patients undergoing arthroscopic RCR. Tear size, number of tendons involved, and subscapularis involvement were significant and additive predictors of suboptimal CSO achievement at 2-year minimum follow-up. Treatment of concurrent biceps pathology with tenodesis conferred 78% increased likelihood of CSO achievement compared with tenotomy.
Keywords: clinically significant outcome; machine learning; patient-reported outcome measurement; rotator cuff repair; rotator cuff tear; shoulder arthroscopy.
© The Author(s) 2025.
Conflict of interest statement
One or more of the authors has declared the following potential conflict of interest or source of funding: E.J.B. has stock or stock options in Pfizer, Inc, and Amgen, Inc. H.H.P. has received hospitality payments from Abbott Laboratories and Medtronic. A.B.Y. has received hospitality payments and consulting fees from Stryker and nonconsulting fees from Arthrex; is a paid consultant for AlloSource, JRF Ortho, Stryker, and Arthrex; has received research support from Arthrex, and Organogenesis; has stock or stock options in Icarus Medical, Patient IQ, and Sparta Biomedical; is an unpaid consultant for Patient IQ and Sparta Biomedical; and is a paid presenter or speaker for Stryker. B.J.C. has received consulting fees from Aesculap Biologics, Arthrex, Bioventus, Enovis, Vericel, OSSIO LTD, Pacira Pharmaceuticals Inc, Endo Pharmaceuticals, and ACUMED LLC; nonconsulting fees from Terumo BCT; IP royalties from Arthrex, and Elsevier Publishing; hospitality payments from GE Healthcare; honoraria from Vericel; research support from Aesculap/B.Braun, Arthrex, and National Institutes of Health (NIAMS and NICHD); and other financial or material support from JRF Ortho. He holds stock or stock options in Bandgrip Inc and Ossio. N.N.V. has received hospitality payments from Abbott Laboratories, Spinal Simplicity, and Relievant Medsystems; royalties from Arthrex, Smith & Nephew, Stryker, and Graymont Professional Products; consulting fees from Arthrex and Stryker; and research support from Breg, Ossur, Smith & Nephew, Stryker, and Arthrex. M.H. has received education payments from Arthrex, Foundation Medical, Medwest Associates, and Smith & Nephew; honoraria from Encore Medical; hospitality payments from Orthalign and Stryker; consulting fees from Enovis, Moximed, and Vericel; a grant from Medical Device Business Services; and publishing royalties and financial or material support from Elsevier. B.F. has received debt forgiveness from Medwest Associates; consulting fees from Stryker and Smith & Nephew; research support from Arthrex, Smith & Nephew, and Stryker; and publishing royalties and financial or material support from Elsevier. He holds stock or stock options in iBrainTech, Sparta Biopharma, and Zuno Medical. AOSSM checks author disclosures against the Open Payments Database (OPD). AOSSM has not conducted an independent investigation on the OPD and disclaims any liability or responsibility relating thereto. Ethical approval for this study was waived by Rush University Medical Center (No. 20041404-IRB01).
Figures
References
-
- Bennett WF. Arthroscopic repair of massive rotator cuff tears: a prospective cohort with 2- to 4-year follow-up. Arthroscopy. 2003;19(4):380-390. - PubMed
-
- Brock G, Pihur V, Datta S, Datta S. ciValid: an R package for cluster validation. J Stat Softw. 2008;25(4):1-22.
-
- Chung SW, Kim JY, Kim MH, Kim SH, Oh JH. Arthroscopic repair of massive rotator cuff tears: outcome and analysis of factors associated with healing failure or poor postoperative function. Am J Sports Med. 2013;41(7):1674-1683. - PubMed
-
- Cvetanovich GL, Gowd AK, Liu JN, et al. Establishing clinically significant outcome after arthroscopic rotator cuff repair. J Shoulder Elbow Surg. 2019;28(5):939-948. - PubMed
LinkOut - more resources
Full Text Sources

