PTX3 as a diagnostic and prognostic biomarker in lung adenocarcinoma: a comprehensive analysis
- PMID: 40537732
- PMCID: PMC12179049
- DOI: 10.1007/s12672-025-02983-5
PTX3 as a diagnostic and prognostic biomarker in lung adenocarcinoma: a comprehensive analysis
Abstract
Background: Lung cancer remains a leading global cause of mortality, with lung adenocarcinoma (LUAD) as the predominant histological subtype. Current serum biomarkers like carcinoembryonic antigen (CEA) lack specificity, necessitating novel diagnostic targets. Pentraxin 3 (PTX3), a homo-multimeric protein downregulated in malignancies, was evaluated for its diagnostic and prognostic roles in LUAD.
Methods: PTX3 expression was analyzed using TCGA/GEO datasets and clinical serum samples (97 LUAD vs. 40 controls). Diagnostic utility was assessed via ROC curves for PTX3, CEACAM5, and their combination. Prognostic value was determined by Kaplan-Meier and Cox regression. PTX3-associated differentially expressed genes (DEGs) were explored through functional enrichment, tumor microenvironment (TME) analysis, and drug sensitivity profiling.
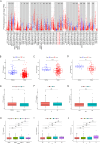
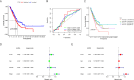
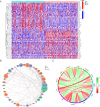
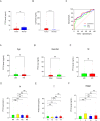
Result: The TCGA and GEO datasets revealed that PTX3 mRNA expression was significantly downregulated in LUAD, and the AUC values with PTX3 were > 0.7. Detection of CEACAM5 and PTX3 combined can improve diagnostic accuracy, and patients with high PTX3 level have shorter overall survival. Multivariate Cox analysis revealed that PTX3 is an independent predictor of overall survival. The result of ELISA further confirmed the low level of PTX3 protein. PTX3 is important in the functional analysis and TME of lung adenocarcinoma. In addition, the sensitivity of tumor cells to anti-cancer drugs is significantly correlated with the expression of PTX3.
Conclusion: PTX3 emerges as a dual biomarker for LUAD diagnosis and prognosis, with mechanistic ties to TME remodeling and therapeutic resistance, highlighting its potential for clinical translation.
Keywords: Diagnosis; Immune; Lung adenocarcinoma; PTX3.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Competing interests: The authors declare no competing interests. Ethics approval and consent to participate: This study was performed in line with the principles of the Declaration of Helsinki. The Ethics Committee of China Medical University People’s Hospital evaluated and approved this work (approval number 2024-K069). Informed consent was obtained from all individual participants included in the study.
Figures


Similar articles
-
Integrative Multi-Omics Analysis and Experimental Validation Identify SPOP as a Prognostic Biomarker and Immune Regulator in Lung Adenocarcinoma.J Cancer. 2025 Jun 23;16(9):2984-2996. doi: 10.7150/jca.111751. eCollection 2025. J Cancer. 2025. PMID: 40657370 Free PMC article.
-
Unraveling the role of GPCR signaling in metabolic reprogramming and immune microenvironment of lung adenocarcinoma: a multi-omics study with experimental validation.Front Immunol. 2025 Jun 6;16:1606125. doi: 10.3389/fimmu.2025.1606125. eCollection 2025. Front Immunol. 2025. PMID: 40547013 Free PMC article.
-
Identification of a coagulation-related signature correlated with immune infiltration and their prognostic implications in lung adenocarcinoma.Thorac Cancer. 2023 Nov;14(33):3295-3308. doi: 10.1111/1759-7714.15121. Epub 2023 Oct 5. Thorac Cancer. 2023. PMID: 37795779 Free PMC article.
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
Cost-effectiveness of using prognostic information to select women with breast cancer for adjuvant systemic therapy.Health Technol Assess. 2006 Sep;10(34):iii-iv, ix-xi, 1-204. doi: 10.3310/hta10340. Health Technol Assess. 2006. PMID: 16959170
References
-
- Siegel RL, Miller KD, Fuchs HE, Jemal A. Cancer statistics, 2022. CA Cancer J Clin. 2022;72(1):7–33. - PubMed
-
- Hendriks LEL, Remon J, Faivre-Finn C, Garassino MC, Heymach JV, Kerr KM, et al. Non-small-cell lung cancer. Nat Rev Dis Primers. 2024;10(1):1–22. - PubMed
-
- Kadara H, Scheet P, Wistuba II, Spira AE. Early events in the molecular pathogenesis of lung Cancer. Cancer Prev Res (Phila). 2016;9(7):518–27. - PubMed
-
- Lee E, Kazerooni EA. Lung Cancer screening. Semin Respir Crit Care Med. 2022;43(6):839–50. - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous
