Characterization and Evaluation of Lactobacillus plantarum LC5.2 Isolated from Thai Native Pigs for its Probiotic Potential in Gut Microbiota Modulation and Immune Enhancement
- PMID: 40537904
- PMCID: PMC12197819
- DOI: 10.4014/jmb.2503.03028
Characterization and Evaluation of Lactobacillus plantarum LC5.2 Isolated from Thai Native Pigs for its Probiotic Potential in Gut Microbiota Modulation and Immune Enhancement
Abstract
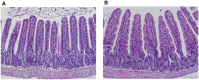
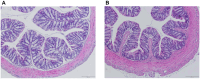
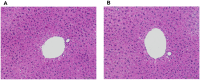
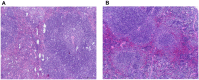
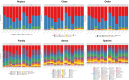
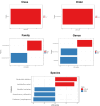
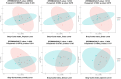
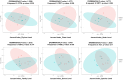
Probiotic supplementation, particularly with Lactobacillus species, enhances growth performance, maintains gastrointestinal microbial balance, and prevents infections in livestock. This study isolated Lactobacillus strains from the feces of healthy native pigs in southern Thailand and assessed their probiotic properties and safety through both in vitro and in vivo evaluations. Nine Lactobacillus strains showed probiotic potential, with Lactobacillus plantarum LC5.2 demonstrating the best characteristics. This strain tolerated both acid and bile (100% tolerance) and exhibited strong adhesion properties, including high auto-aggregation (69.74%), cell surface hydrophobicity (77.14%), adhesion to Caco-2 cells (9.31%), and biofilm formation. It also exhibited antibacterial activity, inhibiting EHEC, EPEC, and Salmonella Typhimurium through organic acid production. Co-aggregation with these pathogens ranged from 60.83% to 74.09%. Safety evaluations showed no hemolytic activity, susceptibility to antibiotics, and co-existence with other probiotics. In mice, L. plantarum LC5.2 showed no toxicity, with normal food intake, behavior, and weight gain. No abnormalities were found in the small intestine, colon, liver, or spleen. Mice administered the probiotic had significantly higher intestinal IgA levels. Gut microbiome analysis revealed no notable structural alterations but indicated an increase in beneficial bacteria, including Lactobacillus. These results suggest that L. plantarum LC5.2 demonstrates strong probiotic potential, safety, and benefits for gut health, making it a promising candidate for livestock applications.
Keywords: Lactobacillus plantarum; Thai native pigs; gut microbiota modulation; probiotic properties; safety assessment.
Conflict of interest statement
The authors have no financial conflicts of interest to declare.
Figures


Similar articles
-
Gut microbiota modulation and immunity enhancement by Bacillus amyloliquefaciens NL1.2: A fiber-degrading probiotic isolated from native Thai swine.Vet World. 2025 Jun;18(6):1487-1507. doi: 10.14202/vetworld.2025.1487-1507. Epub 2025 Jun 10. Vet World. 2025. PMID: 40689169 Free PMC article.
-
A Peek into the Relationship Between Probiotic Characteristics of Lactiplantibacillus plantarum Strains and Sortase A Gene Expression.Curr Microbiol. 2025 Jul 1;82(8):356. doi: 10.1007/s00284-025-04328-5. Curr Microbiol. 2025. PMID: 40591003
-
Lactobacillus plantarum 1-2-3 inhibits ferroptosis by regulating dysregulated fatty acid metabolism to protect mice from high-fat diet-induced MAFLD.Free Radic Biol Med. 2025 Oct;238:137-151. doi: 10.1016/j.freeradbiomed.2025.06.042. Epub 2025 Jun 22. Free Radic Biol Med. 2025. PMID: 40555343
-
Synbiotics, prebiotics and probiotics for solid organ transplant recipients.Cochrane Database Syst Rev. 2022 Sep 20;9(9):CD014804. doi: 10.1002/14651858.CD014804.pub2. Cochrane Database Syst Rev. 2022. PMID: 36126902 Free PMC article.
-
Probiotics for the prevention of pediatric antibiotic-associated diarrhea.Cochrane Database Syst Rev. 2011 Nov 9;(11):CD004827. doi: 10.1002/14651858.CD004827.pub3. Cochrane Database Syst Rev. 2011. Update in: Cochrane Database Syst Rev. 2015 Dec 22;(12):CD004827. doi: 10.1002/14651858.CD004827.pub4. PMID: 22071814 Updated.
References
-
- Zendeboodi F, Khorshidian N, Mortazavian AM, da Cruz AG. Probiotic: conceptualization from a new approach. Curr. Opin. Food Sci. 2020;32:103–123. doi: 10.1016/j.cofs.2020.03.009. - DOI
-
- Anee IJ, Alam S, Begum RA, Shahjahan RM, Khandaker AM. 2021. The role of probiotics on animal health and nutrition. J. Basic Appl. Zool. 82. https://doi.org/10.1186/s41936-021-00250-x. 10.1186/s41936-021-00250-x - DOI
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

