DDAH1 Promotes Cisplatin Chemoresistance in Patients with Locally Advanced Nasopharyngeal Carcinoma via the EGFR-JAK2-STAT3 Pathway
- PMID: 40538138
- PMCID: PMC12376576
- DOI: 10.1002/advs.202503647
DDAH1 Promotes Cisplatin Chemoresistance in Patients with Locally Advanced Nasopharyngeal Carcinoma via the EGFR-JAK2-STAT3 Pathway
Abstract
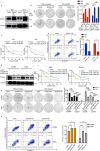
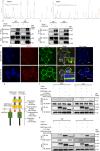
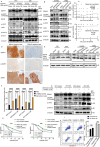
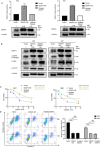
Cisplatin-based induction chemotherapy (IC) improves survival in patients with locally advanced nasopharyngeal carcinoma (LANPC). However, ≈30% of patients with LANPC receiving IC develop chemoresistance, and 20% experience disease progression. The relation between chemoresistance and Dimethylarginine dimethylaminohydrolase-1 (DDAH1) in NPC has not been mentioned in previous studies. To explore the regulatory mechanism and biological function of DDAH1 in cisplatin chemoresistance, NPC cell lines are subjected to overexpression and knockdown of DDAH1 in vitro, with findings further corroborated by in vivo chemosensitivity assays. The predictive value of DDAH1 expression is evaluated for survival and resistance to cisplatin-based IC in a cohort of 339 patients with LANPC. Overexpression of DDAH1 in NPC cell lines increases cisplatin resistance both in vitro and in vivo through binding to the intracellular domain of epidermal growth factor receptor (EGFR), enhancing its dimerization and phosphorylation, thereby promoting the activation of the JAK2-STAT3 pathway, which is dependent on EGFR and extracellular ligands and can be weakened by nimotuzumab. Clinically, DDAH1 positivity correlates with unfavorable 3-year survivals. This study identified DDAH1 as a prognostic marker and a potential therapeutic target for nimotuzumab to overcome treatment failure and chemoresistance in LANPC and other EGFR-positive cancers.
Keywords: DDAH1; EGFR; JAK2‐STAT3 pathway; cisplatin resistance; nasopharyngeal carcinoma; nimotuzumab.
© 2025 The Author(s). Advanced Science published by Wiley‐VCH GmbH.
Conflict of interest statement
The authors declare no conflict of interest.
Figures




Similar articles
-
Role of the CEACAM7-JAK2/STAT3-BST2 Axis in the migration of nasopharyngeal carcinoma cells.Biochim Biophys Acta Mol Basis Dis. 2025 Oct;1871(7):167948. doi: 10.1016/j.bbadis.2025.167948. Epub 2025 Jun 5. Biochim Biophys Acta Mol Basis Dis. 2025. PMID: 40482926
-
The diagnostic and prognostic value of LncRNA FAM3D-AS1 expression in nasopharyngeal carcinoma and its effect on tumor progression and cisplatin resistance.Hereditas. 2025 Aug 26;162(1):171. doi: 10.1186/s41065-025-00526-0. Hereditas. 2025. PMID: 40859288 Free PMC article.
-
AK4 promotes nasopharyngeal carcinoma metastasis and chemoresistance by activating NLRP3 inflammatory complex.Cell Death Dis. 2025 Jul 1;16(1):480. doi: 10.1038/s41419-025-07805-8. Cell Death Dis. 2025. PMID: 40593461 Free PMC article.
-
Erlotinib or gefitinib for the treatment of relapsed platinum pretreated non-small cell lung cancer and ovarian cancer: a systematic review.Drug Resist Updat. 2011 Jun;14(3):177-90. doi: 10.1016/j.drup.2011.02.004. Epub 2011 Mar 24. Drug Resist Updat. 2011. PMID: 21435938
-
Comparison of TPF and PF induction chemotherapy combined with cisplatin concurrent chemoradiotherapy for locoregionally advanced nasopharyngeal carcinoma: A systematic review and meta-analysis.Medicine (Baltimore). 2025 Jan 17;104(3):e41278. doi: 10.1097/MD.0000000000041278. Medicine (Baltimore). 2025. PMID: 39833074 Free PMC article.
References
-
- Wong K. C. W., Hui E. P., Lo K.‐W., Lam W. K. J., Johnson D., Li L., Tao Q., Chan K. C. A., To K.‐F., King A. D., Ma B. B. Y., Chan A. T. C., Nat. Rev. Clin. Oncol. 2021, 18, 679. - PubMed
-
- Chen Y. P., Chan A. T. C., Le Q. T., Blanchard P., Sun Y., Ma J., Lancet 2019, 394, 64. - PubMed
-
- Li K., Lin G.‐Z., Shen J.‐C., Zhou Q., Asian Pac. J. Cancer Prev. 2014, 15, 9899. - PubMed
-
- Sun Y., Li W.‐F., Chen N.‐Y., Zhang N., Hu G.‐Q., Xie F.‐Y., Sun Y., Chen X.‐Z., Li J.‐G., Zhu X.‐D., Hu C.‐S., Xu X.‐Y., Chen Y.‐Y., Hu W.‐H., Guo L., Mo H.‐Y., Chen L., Mao Y.‐P., Sun R., Ai P., Liang S.‐B., Long G.‐X., Zheng B.‐M., Feng X.‐L., Gong X.‐C., Li L., Shen C.‐Y., Xu J.‐Y., Guo Y., Chen Y.‐M., et al., Lancet Oncol. 2016, 17, 1509.
-
- Zhang Y., Chen L., Hu G.‐Q., Zhang N., Zhu X.‐D., Yang K.‐Y., Jin F., Shi M., Chen Y.‐P., Hu W.‐H., Cheng Z.‐B., Wang S.‐Y., Tian Y., Wang X.‐C., Sun Y., Li J.‐G., Li W.‐F., Li Y.‐H., Tang L.‐L., Mao Y.‐P., Zhou G.‐Q., Sun R., Liu X., Guo R., Long G.‐X., Liang S.‐Q., Li L., Huang J., Long J.‐H., Zang J., et al., N. Engl. J. Med. 2019, 381, 1124. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous
