Zileuton protects against arachidonic acid/5-lipoxygenase/leukotriene axis-mediated neuroinflammation in experimental traumatic brain injury
- PMID: 40538546
- PMCID: PMC12176805
- DOI: 10.3389/fphar.2025.1516836
Zileuton protects against arachidonic acid/5-lipoxygenase/leukotriene axis-mediated neuroinflammation in experimental traumatic brain injury
Abstract
Introduction: Traumatic brain injury (TBI) is a leading cause of death and disability globally. Several studies have shown that 5-lipoxygenase (5-LOX) inhibition reduces leukotriene (LT) release and the inflammatory response, attenuating the development of respiratory diseases, myocardial infarction, and ischemic cerebral injury. However, its role in the pathophysiology of TBI remains unclear.
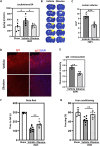
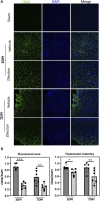
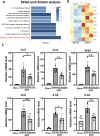
Methods: Controlled cortical impact injury was induced to construct a mouse model of TBI. Pericontusional brain tissue samples from sham and TBI mice at 7 days after injury were used for RNA-seq analysis. Altered gene enrichment following TBI, based on Kyoto Encyclopedia of Genes and Genomes (KEGG) pathway analysis, was quantified through real-time polymerase chain reaction (RT-PCR). Immunocytochemistry, Western blotting, and single-cell sequencing experiments were also performed to analyze 5-Lox protein expression. Arachidonic acid (AA) was detected through liquid chromatography mass spectrometry/mass spectrometry. Enzyme-linked immunosorbent assay was used to detect LTB4 release after TBI with or without zileuton treatment. Brain damage, blood-brain barrier disruption, and neuronal apoptosis were detected through histological examination. Neurological outcomes were determined through rotarod and fear conditioning tests.
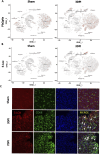
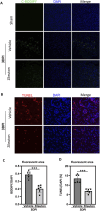
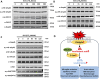
Results: TBI induced significant upregulation of genes related to the AA metabolic pathway, particularly the AA/5-LOX/LT axis, as verified by RT-PCR. AA and LTB4 production increased significantly after TBI. The expression levels of Pla2g4a, which hydrolyses phospholipids to release AA, and 5-Lox, which in turn act downstream to convert AA to LT, were dramatically upregulated up to 7 days after TBI. 5-LOX accumulated in the cytoplasm of activated ameboid microglial cells. In vivo, 5-LOX inhibition with zileuton blocked LT release and reduced microglial activation and the production of inflammatory cytokines, including Il-1β, Ccl7, Spp1, Ccr1, Ccl2, and Il-10. Zileuton also reduced TBI-induced lipid ROS and neuronal cell apoptosis, ameliorating brain damage compared to the vehicle group and improving neurological outcomes after TBI. Mechanically, TBI-induced LT upregulation may stimulate BV2 microglial activation through the ERK, NF-κB, and Akt pathways.
Conclusion: Our findings demonstrated the role of 5-LOX in TBI and its potential as a therapeutic target in TBI treatment.
Keywords: 5-lipoxygenase; leukotriene; microglial cells; neuroinflammation; traumatic brain injury; zileuton.
Copyright © 2025 Xu, Wang, Li, Yang, Yang, Tang, Lu and Yuan.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

References
-
- Attaluri S., Upadhya R., Kodali M., Madhu L. N., Upadhya D., Shuai B., et al. (2022). Brain-specific increase in leukotriene signaling accompanies chronic neuroinflammation and cognitive impairment in a model of Gulf war illness. Front. Immunol. 13, 853000. 10.3389/fimmu.2022.853000 - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

