A stress-sensing circuit signals to the central pacemaker to reprogram circadian rhythms
- PMID: 40540570
- PMCID: PMC12180509
- DOI: 10.1126/sciadv.adr7960
A stress-sensing circuit signals to the central pacemaker to reprogram circadian rhythms
Abstract
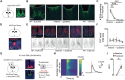
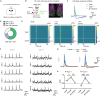
The circadian system provides a temporal framework for animals to anticipate environmental events, including threats. However, the effects of stressors on the circadian system remain poorly understood. Here, we demonstrate that, in mice, stressors shift the phase of the central pacemaker, housed in the suprachiasmatic nucleus (SCN), through glutamatergic inputs from the anterior paraventricular nucleus of the thalamus (aPVT). Unlike light, which can phase delay or advance the central pacemaker, stressors consistently induce delays, effects attenuated by inhibiting aPVT neurons. Stressors robustly activate AVP-expressing neurons within the SCN and are associated with inhibition of VIP-expressing neurons, whereas light strongly activates VIP-expressing neurons with minimal effects on AVP-expressing neurons. Pairing stressors with light reveals distinct time-dependent interactions, enhancing phase delays at early night but abolishing phase advances at late night. Our findings uncover distinct SCN microcircuits that differentially encode light and stressors, providing insights into how environmental cues modulate circadian timing.
Figures






Similar articles
-
Functional Roles of Gastrin-Releasing Peptide-Producing Neurons in the Suprachiasmatic Nucleus: Insights into Photic Entrainment and Circadian Regulation.J Neurosci. 2025 Jun 18;45(25):e0065252025. doi: 10.1523/JNEUROSCI.0065-25.2025. J Neurosci. 2025. PMID: 40404352 Free PMC article.
-
Computational modeling of the synergistic role of GCN2 and the HPA axis in regulating the integrated stress response in the central circadian timing system.Physiol Genomics. 2024 Aug 1;56(8):531-543. doi: 10.1152/physiolgenomics.00030.2024. Epub 2024 Jun 17. Physiol Genomics. 2024. PMID: 38881429 Free PMC article.
-
Mitofusin 2 controls mitochondrial and synaptic dynamics of suprachiasmatic VIP neurons and related circadian rhythms.J Clin Invest. 2025 Jul 1;135(13):e185000. doi: 10.1172/JCI185000. eCollection 2025 Jul 1. J Clin Invest. 2025. PMID: 40590229 Free PMC article.
-
An essential role for peptidergic signalling in the control of circadian rhythms in the suprachiasmatic nuclei.J Neuroendocrinol. 2003 Apr;15(4):335-8. doi: 10.1046/j.1365-2826.2003.01005.x. J Neuroendocrinol. 2003. PMID: 12622830 Review.
-
Neuroglial and synaptic rearrangements associated with photic entrainment of the circadian clock in the suprachiasmatic nucleus.Eur J Neurosci. 2010 Dec;32(12):2133-42. doi: 10.1111/j.1460-9568.2010.07520.x. Eur J Neurosci. 2010. PMID: 21143667 Review.
References
-
- Hut R. A., Kronfeld-Schor N., van der Vinne V., De la Iglesia H., In search of a temporal niche: Environmental factors. Prog. Brain Res. 199, 281–304 (2012). - PubMed
-
- De Kloet E. R., Joëls M., Holsboer F., Stress and the brain: From adaptation to disease. Nat. Rev. Neurosci. 6, 463–475 (2005). - PubMed
-
- American Psychiatric Association, Diagnostic and Statistical Manual of Mental Disorders: DSM-5 (American Psychiatric Association, 2013), vol. 5.
-
- Cain S. W., Verwey M., Hood S., Leknickas P., Karatsoreos I., Yeomans J. S., Ralph M. R., Reward and aversive stimuli produce similar nonphotic phase shifts. Behav. Neurosci. 118, 131–137 (2004). - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

