The effects of extracorporeal blood purification (oXiris®) in patients with cardiogenic shock who require VA-ECMO (CLEAN ECMO): a prospective, open-label, randomized controlled pilot study
- PMID: 40542431
- PMCID: PMC12181899
- DOI: 10.1186/s13054-025-05495-4
The effects of extracorporeal blood purification (oXiris®) in patients with cardiogenic shock who require VA-ECMO (CLEAN ECMO): a prospective, open-label, randomized controlled pilot study
Abstract
Background: A systemic inflammatory response can contribute to poor outcomes in an advanced stage of cardiogenic shock (CS). We investigated the efficacy of extracorporeal endotoxin and cytokine adsorption using oXiris in patients with CS undergoing venoarterial extracorporeal membrane oxygenation (VA-ECMO).
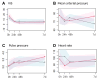
Methods: In this prospective, single-center, randomized, open-label pilot trial, 40 patients with CS who were undergoing VA-ECMO were randomly assigned to receive either oXiris for 24 h (n = 20) or usual care (n = 20). The primary endpoint was endotoxin levels at 48 h. Secondary endpoints included changes in inflammatory cytokines, vasoactive-inotropic score (VIS), ECMO weaning success, and in-hospital and 30-day mortality.
Results: The median endotoxin levels at 48 h were 0.5 (IQR 0.4-1.0) in the oXiris group and 0.4 (IQR 0.2-0.5) in the control group, with no significant difference between them (P = 0.097). The oXiris group showed significant temporal reductions in GDF-15 and IL-6 levels, with IL-6 revealing significant reductions from baseline to 24 h (P = 0.020) and from baseline to 7 days (P = 0.003). VIS decreased significantly from baseline to 48 h (-13.63, 95% CI: -20.90 - -6.34, P < 0.001) and 7 days (-12.19, 95% CI: -21.0 - -3.31, P = 0.007) in the oXiris group, but intergroup differences were insignificant. ECMO weaning success, duration of ECMO support, and mortality rates were similar between the groups.
Conclusion: In this pilot study conducted on CS patients requiring VA-ECMO, oXiris treatment did not significantly reduce endotoxin levels or improve patient centered clinical outcomes.
Trial registration: NCT05642273, registered 8 December 2022.
Keywords: Cardiogenic shock; Cytokine; Endotoxin; Extracorporeal membrane oxygenation; Inflammatory marker.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: The research was conducted according to the Declaration of Helsinki, and the study protocol was approved by the institutional review board of Samsung Medical Center (No. 2022-09-082) and registered at ClinicalTrials.gov (NCT05642273, registered 8 December 2022). All participants or their representatives provided written informed consent to participate. Consent for publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures


References
-
- van Diepen S, Katz JN, Albert NM, Henry TD, Jacobs AK, Kapur NK, et al. Contemporary management of cardiogenic shock: A scientific statement from the American heart association. Circulation. 2017;136(16):e232–68. - PubMed
-
- Rob D, Belohlavek J. The mechanical support of cardiogenic shock. Curr Opin Crit Care. 2021;27(4):440–6. - PubMed
-
- Kohsaka S, Menon V, Lowe AM, Lange M, Dzavik V, Sleeper LA, et al. Systemic inflammatory response syndrome after acute myocardial infarction complicated by cardiogenic shock. Arch Intern Med. 2005;165(14):1643–50. - PubMed
-
- Adrie C, Adib-Conquy M, Laurent I, Monchi M, Vinsonneau C, Fitting C, et al. Successful cardiopulmonary resuscitation after cardiac arrest as a sepsis-like syndrome. Circulation. 2002;106(5):562–8. - PubMed
-
- Geppert A, Dorninger A, Delle-Karth G, Zorn G, Heinz G, Huber K. Plasma concentrations of interleukin-6, organ failure, vasopressor support, and successful coronary revascularization in predicting 30-day mortality of patients with cardiogenic shock complicating acute myocardial infarction. Crit Care Med. 2006;34(8):2035–42. - PubMed
Publication types
MeSH terms
Substances
Associated data
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

