Combinational Therapy of Mesenchymal Stem Cells and Metformin in Bleomycin-Induced Idiopathic Pulmonary Fibrosis in Rat Model
- PMID: 40542974
- PMCID: PMC12334527
- DOI: 10.1007/s12010-025-05289-y
Combinational Therapy of Mesenchymal Stem Cells and Metformin in Bleomycin-Induced Idiopathic Pulmonary Fibrosis in Rat Model
Abstract
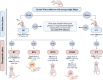
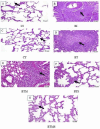
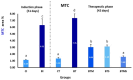
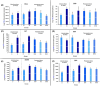
Idiopathic pulmonary fibrosis (IPF) is a progressive and severe lung disease characterized by the buildup of interstitial fibrosis, where excessive collagen accumulates, leading to airway obstruction. This condition is initiated by the abnormal proliferation of alveolar type II (AT2) cells. Metformin, an established antidiabetic drug, has gained attention for its repurposed use as an anti-fibrotic agent. Meanwhile, adipose-derived mesenchymal stem cells (ADMSCs) exhibit potent anti-inflammatory and regenerative properties, and they have been shown to reduce collagen deposition. In this study, we hypothesize that the combination of metformin and ADMSCs can synergistically alleviate IPF and promote healthy lung tissue regeneration in a rat model. The goal is to evaluate the safety and efficacy of this approach at multiple levels; biochemical, molecular, histopathological, and histochemical. To induce IPF, Wistar albino rats received a single intratracheal dose of bleomycin (5 mg/kg body weight). The therapeutic phase involved treatment with either metformin or ADMSCs or a combination of both. Metformin was administered intraperitoneally (65 mg/kg body weight) every other day, while ADMSCs were delivered intravenously (1 × 10⁶ cells/0.5 ml DMEM/rat) through the tail vein. Our results demonstrated the effectiveness of combinational therapy, especially in mitigating oxidative stress. This was evidenced by the restoration of oxidative stress biomarkers, malondialdehyde (MDA), and catalase (CAT), as well as the regulation of collagenase type IV (MMP9), bovine serum albumin (BSA), and total protein levels in lung tissues. Moreover, the therapy modulated the expression of key inflammatory and fibrotic genes, including the pro-fibrotic marker TGF-β1, proinflammatory cytokine IL-6, and anti-inflammatory cytokine IL-10. Histopathological and histochemical analyses further supported the therapeutic benefits, showing significant recovery from bleomycin-induced fibrosis in rats treated with either the single or combined therapy. The findings suggest that this combinational approach could be a promising strategy for IPF treatment by simultaneously reducing inflammation, oxidative stress, and fibrosis while promoting tissue regeneration.
Keywords: Bleomycin; Collagen deposition; Idiopathic pulmonary fibrosis; Mesenchymal stem cells; Metformin.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics Approval: Experimental protocols and procedures used in this study were approved by the Cairo University Institutional Animal Care and Use Committee (CU-IACUC) (Egypt), (approval no. CU/I/F/10/24) in accordance with the international guidelines for care and use of laboratory animals. Consent to Participate: Not applicable. Consent for Publication: Not applicable. Competing interests: The authors declare no competing interests.
Figures


Similar articles
-
Adipose-derived mesenchymal stem cell therapy for reverse bleomycin-induced experimental pulmonary fibrosis.Sci Rep. 2023 Aug 14;13(1):13183. doi: 10.1038/s41598-023-40531-9. Sci Rep. 2023. PMID: 37580529 Free PMC article.
-
Codonopsis radix improves bleomycin-induced idiopathic pulmonary fibrosis by regulating TGF-β1/Smad signaling pathway and inhibiting fibroblast differentiation and proliferation.J Ethnopharmacol. 2025 Aug 29;352:120233. doi: 10.1016/j.jep.2025.120233. Epub 2025 Jul 5. J Ethnopharmacol. 2025. PMID: 40619039
-
Human bone marrow mesenchymal stem cell-derived extracellular vesicles induce inverse dose-dependent anti-fibrotic effects in human myofibroblast cultures and bleomycin-injured mice with pulmonary fibrosis.Biomed Pharmacother. 2025 Sep;190:118370. doi: 10.1016/j.biopha.2025.118370. Epub 2025 Jul 21. Biomed Pharmacother. 2025. PMID: 40695046
-
Effectivity of mesenchymal stem cells for bleomycin-induced pulmonary fibrosis: a systematic review and implication for clinical application.Stem Cell Res Ther. 2021 Aug 23;12(1):470. doi: 10.1186/s13287-021-02551-y. Stem Cell Res Ther. 2021. PMID: 34420515 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
References
-
- Martinez, F. J., Collard, H. R., Pardo, A., Raghu, G., Richeldi, L., Selman, M., … Wells, A. U. (2017). Idiopathic pulmonary fibrosis. Nature Reviews Disease Primers, 3(1), 17074. 10.1038/nrdp.2017.74 - PubMed
-
- Molina-Molina, M., Aburto, M., Acosta, O., Ancochea, J., Rodríguez-Portal, J. A., Sauleda, J., … Xaubet, A. (2018). Importance of early diagnosis and treatment in idiopathic pulmonary fibrosis. Expert Review of Respiratory Medicine, 12(7), 537–539. 10.1080/17476348.2018.1472580 - PubMed
-
- Koudstaal, T., & Wijsenbeek, M. S. (2023). Idiopathic pulmonary fibrosis. La Presse Médicale,52(3), Article 104166. 10.1016/j.lpm.2023.104166 - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Miscellaneous

