A novel approach to enhance glioblastoma multiforme treatment efficacy: non-coding RNA targeted therapy and adjuvant approaches
- PMID: 40544306
- PMCID: PMC12181852
- DOI: 10.1186/s13148-025-01900-5
A novel approach to enhance glioblastoma multiforme treatment efficacy: non-coding RNA targeted therapy and adjuvant approaches
Abstract
Background: Glioblastoma multiforme (GBM) is a lethal brain tumor. With the current gold standard chemotherapy treatment, temozolomide (TMZ), many patients do not survive beyond one year. While the urgency of researching novel treatments is understandable, the prohibitively high costs and the prolonged duration of research and clinical trials significantly delay the availability of medical advancements to the general public. This highlights the urgent need for adjuvant therapies to enhance treatment effectiveness.
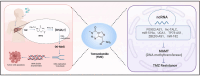
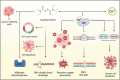
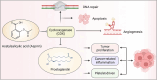
Main body: Recent research has suggested the potential of repurposing FDA-approved drugs such as temozolomide (TMZ), disulfiram (DSF), and aspirin for the treatment of glioblastoma, with encouraging evidence particularly for DSF and aspirin. Additionally, compounds like histone deacetylase inhibitors (e.g., vorinostat) are being investigated for their impact on non-coding RNA (ncRNA) modulation, including microRNAs (miRNAs) and long non-coding RNAs (lncRNAs). Combining traditional therapies with ncRNA modulation has shown potential in enhancing therapeutic efficacy and targeting specificity. NcRNAs play a crucial role in regulating gene expression and have been implicated in tumor growth, invasion, and treatment resistance. Recent discoveries, such as cuproptosis, offer new insights into tumor cell death mechanisms.
Conclusion: This review focuses on how these molecular insights can serve as novel therapeutic targets and how drug adjuvant therapy may improve GBM treatment strategies. It focuses on how the integration of ncRNA modulation with conventional therapies and the combination strategy of enhancing efficacy of drugs can enhance treatment efficacy and pave the way for innovative approaches in managing GBM. In short, we will explore how non-coding RNAs (ncRNAs) might serve as promising targets and how repurposing TMZ, DSF, and aspirin could help enhance the efficacy of GBM treatment.
Keywords: Aspirin; Cuproptosis; Disulfiram; Glioblastoma multiforme; Non-coding RNA; Temozolomide.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Availability of data and material: No datasets were generated or analyzed during the current study. Ethical approval and consent to participate: Not applicable Consent for publication: We grant our consent for the publication of this work, acknowledging that all authors have reviewed and approved the final manuscript. Competing interests: The authors declare no competing interests.
Figures

References
-
- Wirsching HG, Weller M. Glioblastoma. Malig Brain Tumors State-of-the-Art Treat. 2017;265–288.
-
- Verhaak RG, Hoadley KA, Purdom E, Wang V, Qi Y, Wilkerson MD, Miller CR, Ding L, Golub T, Mesirov JP. Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1[J]. Cancer Cell. 2010;17(1):98–110. - DOI - PMC - PubMed
Publication types
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

