Notch3 enhances the synergistic effect of all-trans retinoic acid and calcipotriol in pancreatic stellate cell activation
- PMID: 40545532
- PMCID: PMC12183861
- DOI: 10.1186/s12967-025-06666-1
Notch3 enhances the synergistic effect of all-trans retinoic acid and calcipotriol in pancreatic stellate cell activation
Abstract
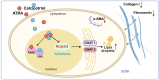
Background: Chronic pancreatitis (CP) is characterized by progressive fibrosis and the activation of pancreatic stellate cells (PSCs). As major producers of collagen-I and fibronectin, PSCs play important roles in pancreatic fibrosis, but few studies have explored methods to target activated PSCs. Notch3, a receptor in the Notch signaling pathway, is highly expressed in activated PSCs, but its specific effect on PSC activation needs to be confirmed. All-trans retinoic acid (ATRA) and the vitamin D analog calcipotriol were able to influence the activation of PSCs, but the relationship between ATRA, calcipotriol and Notch3 has not yet been clarified, and the effects of ATRA and calcipotriol on PSC activation need to be further enhanced.
Methods: The impact of Notch3 on pancreatic stellate cell (PSC) activation was evaluated by knocking down Notch3 in PSCs. PSCs were incubated with ATRA and calcipotriol individually or in combination to explore their effects on PSC activation. Notch3-knockdown PSCs were treated with ATRA or calcipotriol under various conditions in vitro to assess their effects on activation. Nuclear receptor inhibition was used to dissect the interplay between Notch3 signaling and ATRA/calcipotriol pathways. The roles of Notch3, ATRA, and calcipotriol were investigated in vivo using a chronic pancreatitis model. Different combinations of these interventions were tested in the chronic pancreatitis model to evaluate their in vivo efficacy.
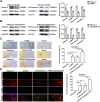
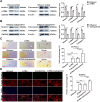
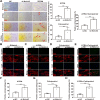
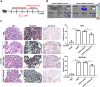
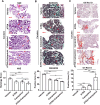
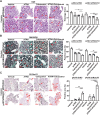
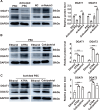
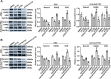
Results: In this study, we confirmed the important role of Notch3 in PSC activation and found that ATRA and calcipotriol could regulate Notch3 expression. Furthermore, that ATRA and calcipotriol can synergistically prevent and reverse PSC activation, whereas knockdown of Notch3 can enhanced this synergistic effect. In CP model, we verified the effect of targeting Notch3 in combination with ATRA and calcipotriol. At last, we found that ATRA and calcipotriol do not regulate Notch3 through the nuclear receptors RARβ and VDR, but ATRA and calcipotriol depend on Notch3 to regulate PSC activation.
Conclusions: Notch3 is a target for inhibiting PSC activation, ATRA and calcipotriol regulate Notch3 expression in PSCs, and targeting Notch3 in combination with ATRA and calcipotriol against PSC activation holds promise as a novel therapeutic approach for treating pancreatic fibrosis in CP.
Keywords: All-trans retinoic acid; Calcipotriol; Chronic pancreatitis; Fibrosis; Notch3; Pancreatic stellate cells.
© 2025. The Author(s).
Conflict of interest statement
Declarations. Ethics approval and consent to participate: All animal experiments are conduct following ethical policies and procedures approved by the Capital Medical University ethics committee (Ethical inspection No. AEEI-2015-143). Consent for publication: Not applicable. Competing interests: The authors report no competing interests.
Figures

Similar articles
-
COMP promotes pancreatic fibrosis by activating pancreatic stellate cells through CD36-ERK/AKT signaling pathways.Cell Signal. 2024 Jun;118:111135. doi: 10.1016/j.cellsig.2024.111135. Epub 2024 Mar 11. Cell Signal. 2024. PMID: 38479555
-
Calpain Inhibitor Calpeptin Improves Pancreatic Fibrosis in Mice with Chronic Pancreatitis by Inhibiting the Activation of Pancreatic Stellate Cells.Curr Mol Pharmacol. 2024;17:e18761429241425. doi: 10.2174/0118761429241425231107044453. Curr Mol Pharmacol. 2024. PMID: 38258594
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of paclitaxel, docetaxel, gemcitabine and vinorelbine in non-small-cell lung cancer.Health Technol Assess. 2001;5(32):1-195. doi: 10.3310/hta5320. Health Technol Assess. 2001. PMID: 12065068
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
References
-
- Xiao AY, Tan ML, Wu LM, Asrani VM, Windsor JA, Yadav D, Petrov MS. Global incidence and mortality of pancreatic diseases: a systematic review, meta-analysis, and meta-regression of population-based cohort studies. Lancet Gastroenterol Hepatol. 2016;1(1):45–55. - PubMed
-
- Vege SS, Chari ST. Chronic pancreatitis. N Engl J Med. 2022;386(9):869–78. - PubMed
-
- Beyer G, Habtezion A, Werner J, Lerch MM, Mayerle J. Chronic pancreatitis. Lancet (London, England). 2020;396(10249):499–512. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

