Buyang Huanwu Decoction Modulates the Gut Microbiota-C/EBPβ/AEP Axis to Ameliorate Cognitive Impairment in Alzheimer's Disease Mice
- PMID: 40546239
- PMCID: PMC12183522
- DOI: 10.1111/cns.70480
Buyang Huanwu Decoction Modulates the Gut Microbiota-C/EBPβ/AEP Axis to Ameliorate Cognitive Impairment in Alzheimer's Disease Mice
Abstract
Background: Alzheimer's disease (AD) is a progressive neurodegenerative disorder characterized by cognitive decline and behavioral disturbances. Buyang Huanwu Decoction (BYHWD), a traditional Chinese herbal formulation, has demonstrated potential neuroprotective effects. This study aims to evaluate the therapeutic impact of BYHWD on cognitive impairments in 3×Tg mice and to investigate its underlying mechanism through modulation of the gut microbiota-C/EBPβ/AEP signaling pathway.
Methods: In two independent experiments, we assessed the effects of BYHWD and its derived fecal microbiota transplantation (FMT-BYHWD) on behavioral performance, neuropathological alterations, and signaling pathways in 3×Tg mice.
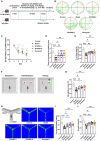
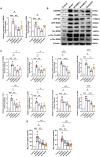
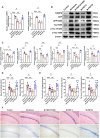
Results: Treatment with BYHWD significantly improved cognitive function in 3×Tg mice and mitigated AD-like pathological changes. By suppressing the C/EBPβ/AEP signaling pathway, BYHWD reduced pathological Aβ plaque deposition, diminished tau hyperphosphorylation, and inhibited the release of pro-inflammatory cytokines. Further analysis revealed that BYHWD restored gut microbiota balance and suppressed the activation of the C/EBPβ/AEP pathway in the hippocampus. Moreover, transplanting FMT-BYHWD from BYHWD-treated mice to germ-free 3×Tg mice also ameliorated their cognitive deficits and AD-like pathology, suggesting that the anti-AD effects of BYHWD are mediated through the gut-brain axis by regulating the interplay between gut microbiota and the C/EBPβ/AEP signaling pathway.
Conclusion: This study uncovers the mechanism by which BYHWD improves cognitive deficits and neuropathological changes in 3×Tg mice via the gut-brain axis, mediated by the modulation of the gut microbiota-C/EBPβ/AEP signaling pathway, providing a novel therapeutic strategy for AD.
Keywords: AEP; Alzheimers disease; Buyang Huanwu decoction; C/EBPβ; amyloid‐β; gut microbiota.
© 2025 The Author(s). CNS Neuroscience & Therapeutics published by John Wiley & Sons Ltd.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



References
-
- Lauwers E., Lalli G., Brandner S., et al., “Potential Human Transmission of Amyloid β Pathology: Surveillance and Risks,” Lancet Neurology 19, no. 10 (2020): 872–878. - PubMed
-
- Collins S. M., Surette M., and Bercik P., “The Interplay Between the Intestinal Microbiota and the Brain,” Nature Reviews Microbiology 10, no. 11 (2012): 735–742. - PubMed
-
- van der Kant R., Goldstein L. S. B., and Ossenkoppele R., “Amyloid‐β‐Independent Regulators of Tau Pathology in Alzheimer Disease,” Nature Reviews Neuroscience 21, no. 1 (2020): 21–35. - PubMed
-
- Junyi L., Yueyang W., Bin L., et al., “Gut Microbiota Mediates Neuroinflammation in Alzheimer's Disease: Unraveling Key Factors and Mechanistic Insights,” Molecular Neurobiology 62, no. 3 (2025): 3746–3763. - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

