Enitociclib, a selective CDK9 inhibitor: in vitro and in vivo preclinical studies in multiple myeloma
- PMID: 40546728
- PMCID: PMC12182852
- DOI: 10.1016/j.bneo.2024.100050
Enitociclib, a selective CDK9 inhibitor: in vitro and in vivo preclinical studies in multiple myeloma
Abstract
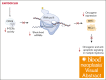
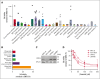
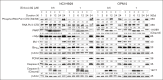
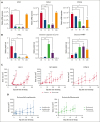
Multiple myeloma (MM) is a cancer of plasma cells that remains incurable despite advances in treatment options. In this study, a library of 216 clinically feasible small-molecule inhibitors was screened to identify agents that selectively inhibit MM cell proliferation. Enitociclib, a cyclin-dependent kinase 9-specific small-molecule inhibitor, was found to be highly effective in decreasing cell viability and inducing apoptosis in 4 MM cell lines. Enitociclib inhibited the phosphorylation of the carboxy-terminal domain (CTD) of RNA polymerase II at Ser2/Ser5 and repressed the protein expression of oncogenes c-Myc, myeloid cell leukemia-1 (Mcl-1), and proliferating cell nuclear antigen (PCNA) in MM cells. Additionally, enitociclib demonstrated synergistic effects with several anti-MM agents, including bortezomib, lenalidomide, pomalidomide, and venetoclax. These results suggest that enitociclib may represent a promising therapeutic option for the treatment of MM, either as a single agent or in combination with other anti-MM agents.
© 2025 by The American Society of Hematology. Licensed under Creative Commons Attribution-NonCommercial-NoDerivatives 4.0 International (CC BY-NC-ND 4.0), permitting only noncommercial, nonderivative use with attribution. All other rights reserved.
Conflict of interest statement
Conflict-of-interest disclosure: M.M.F., B.S.-L., A.J.J., J.B., R.I., and A.H. report employment with Vincerx Pharma. The remaining authors declare no competing financial interests.
Figures


References
-
- Palumbo A, Anderson K. Multiple myeloma. N Engl J Med. 2011;364(11):1046–1060. - PubMed
-
- Diamond JR, Boni V, Lim E, et al. First-in-human dose-escalation study of cyclin-dependent kinase 9 inhibitor VIP152 in patients with advanced malignancies shows early signs of clinical efficacy. Clin Cancer Res. 2022;28(7):1285–1293. - PubMed
LinkOut - more resources
Full Text Sources
Miscellaneous

