Synthesis of novel derivatives from machilin C/D as antiproliferative agents against triple negative breast cancer
- PMID: 40547234
- PMCID: PMC12180578
- DOI: 10.1016/j.ejmcr.2025.100272
Synthesis of novel derivatives from machilin C/D as antiproliferative agents against triple negative breast cancer
Abstract
Lignans are small polyphenolic compounds that play an active role in plant defense against pathogens and predators. Recently, lignan machilin D was reported to be effective as an anti-tumorigenic agent in triple negative breast cancer (TNBC) tumor-bearing mice. Previous studies have relied on plant-extracted material limiting scalability and diversification of the natural scaffold. Herein, we describe a generalizable one-pot synthesis of machilin D and its derivatives via an iron chloride-induced dimerization of isoeugenol. Employing this synthetic methodology allowed for a robust diversification campaign to access seven (7) new lignan derivatives of the machilin D family with superior anti-proliferative properties in 2D and 3D TNBC models. Overall, this work enables lignan natural product-based drug discovery as a platform to identify new probes to elucidate lignan targets in biology and therapeutics for aggressive cancers such as TNBC.
Keywords: Antiproliferatives; Dehydrodiisoeugenol; Iron chloride; Isoeugenol; Licarin A; Lignans; Machilin C; Machilin D; Machilus thunbergii; Natural products; Saururus chinensis; Traditional Chinese medicine; Triple negative breast cancer.
Conflict of interest statement
The authors declare the following competing financial interest(s): A. A., B.C.L. and S.G.A. have patents pending to the University of Kentucky Research Foundation. Declaration of competing interest The authors declare the following competing financial interest(s): The authors declare the following financial interests/personal relationships which may be considered as potential competing interests: Samuel G. Awuah has patents pending to University of Kentucky Research Foundation.
Figures

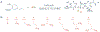


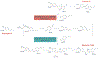
References
-
- Gang DR, et al. , Regiochemical control of monolignol radical coupling: a new paradigm for lignin and lignan biosynthesis, Chem. Biol 6 (3) (1999) 143–151. - PubMed
-
- Dzobo K, The role of natural products as sources of therapeutic agents for innovative drug discovery, Comprehensive Pharmacology (2022) 408–422.
Grants and funding
LinkOut - more resources
Full Text Sources
