Cladribine tablets in the new multiple sclerosis era
- PMID: 40547863
- PMCID: PMC12179467
- DOI: 10.1177/17562864251342855
Cladribine tablets in the new multiple sclerosis era
Abstract
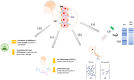
Multiple sclerosis (MS) is an autoimmune condition characterized by inflammatory demyelination that leads to irreversible neurological damage within the central nervous system (CNS). This review examines the therapeutic potential and clinical efficacy of cladribine tablets (CladT) for treating MS, focusing on the immune reconstitution mechanism and CNS effects. CladT represents a notable advance among disease-modifying therapies for MS due to its selective targeting of lymphocytes, resulting in sustained yet reversible immune modulation. This action leads to a substantial reduction in memory B cells while preserving the innate immune system and maintaining immunoglobulin levels, thereby mitigating the risks of secondary autoimmunity and infection. Cladribine appears to penetrate the blood-brain barrier, as indicated by cerebrospinal fluid (CSF) studies from parenteral cladribine use. In MS, CladT is associated with reductions in CSF immunological markers, such as oligoclonal bands and neurofilament light chain levels; it also mitigates acute and chronic inflammation, as evidenced, respectively, by consistent reductions in unique active lesions, and significant decrease in slowly expanding lesions in patients with predominant grey matter damage. These findings underscore the potential of CladT in reducing disability accumulation and improving long-term clinical outcomes in patients with highly active disease. By synthesizing data from clinical trials and real-world studies, this review underscores the effectiveness of CladT in reducing relapse rates, disability progression and magnetic resonance imaging-detected disease activity and emphasizes the importance of early high-efficacy treatment for optimizing long-term outcomes. Furthermore, emerging biomarkers are discussed as potential tools for predicting individual responses to therapy, thereby enabling more personalized treatment strategies. This review also provides valuable insights into the positive impact of CladT on quality of life measures, long-term outcomes and safety profile, all of which support the use of CladT in the evolving landscape of MS management.
Keywords: B cells; T cells; cladribine tablets; disease-modifying therapy; multiple sclerosis.
Plain language summary
Cladribine tablets: a breakthrough in multiple sclerosis treatment This review examines the effectiveness of cladribine tablets (CladT) as a treatment for multiple sclerosis (MS), a chronic autoimmune disease that damages the central nervous system. The article discusses how CladT works by selectively targeting specific immune cells, reducing harmful memory B cells while preserving the overall immune system’s function. Notably, CladT can cross the blood-brain barrier, help reduce key markers of MS, and mitigate acute and chronic inflammation in the brain. Evidence from clinical trials and real-world studies demonstrates that CladT significantly decreases relapse rates and slows disability progression, suggesting that early treatment with CladT can lead to improved long-term outcomes. Additionally, the review explores potential biomarkers that could personalize MS treatment and highlights how CladT’s benefits and safety contribute to improving the quality of life for people with MS.
© The Author(s), 2025.
Figures


References
-
- Bø L, Vedeler CA, Nyland HI, et al. Subpial demyelination in the cerebral cortex of multiple sclerosis patients. J Neuropathol Exp Neurol 2003; 62: 723–732. - PubMed
-
- Kutzelnigg A, Lucchinetti CF, Stadelmann C, et al. Cortical demyelination and diffuse white matter injury in multiple sclerosis. Brain 2005; 128: 2705–2712. - PubMed
Publication types
LinkOut - more resources
Full Text Sources

