Three-Dimensional Culture of Orbital Fibroblasts From Thyroid Eye Disease Induce In Vivo-Like Tissue Remodeling and Fibrosis
- PMID: 40548633
- PMCID: PMC12186838
- DOI: 10.1167/iovs.66.6.67
Three-Dimensional Culture of Orbital Fibroblasts From Thyroid Eye Disease Induce In Vivo-Like Tissue Remodeling and Fibrosis
Abstract
Purpose: This study aimed to investigate the characteristics and molecular mechanisms of orbital fibroblasts under three-dimensional (3D)-culture conditions.
Methods: Orbital connective tissue was collected from patients with thyroid eye disease (TED) and normal controls. Primary fibroblasts were cultured and used to generate 3D microspheres via the hanging drop. These spheroids were cultured for nine days, followed by biomechanical testing, transmission electron microscopy (TEM), and RNA sequencing for transcriptomic analysis. Multiplex immunofluorescence staining was used to assess fibrosis markers, and quantitative PCR validated gene expression changes. TED and normal control (NC) tissues, as well as primary cultured fibroblasts, were also subjected to transcriptomic sequencing.
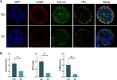
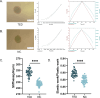
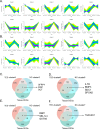
Results: TED-3D microspheres exhibited enhanced contractility, denser fiber deposition, and a characteristic fibrous ring at the periphery. TEM revealed more extracellular matrix (ECM) deposition and stronger tissue remodeling in TED-3D. Fibrosis markers (α-SMA, COL1A1, FN1) increased significantly in TED-3D. Biomechanical testing showed higher stiffness in TED-3D compared to NC-3D. Transcriptomic analysis revealed significant differences, with genes involved in ECM remodeling and fibrosis pathways enriched in TED-3D. Transcriptomic comparison of TED-tissue, TED-2D, and TED-3D revealed that TED-3D is closer to tissue than TED-2D.
Conclusions: The 3D culture of orbital fibroblasts from TED induces in vivo-like tissue remodeling and fibrosis features. Compared to traditional two-dimensional culture, the expression pattern of TED-3D is closer to tissue, making it a more effective model for studying the mechanisms of TED-related fibrosis.
Conflict of interest statement
Disclosure:
Figures






References
-
- Lazarus JH. Epidemiology of Graves’ orbitopathy (GO) and relationship with thyroid disease. Best Pract Res Clin Endocrinol Metab. 2012; 26: 273–279. - PubMed
-
- Taylor PN, Zhang L, Lee RWJ, et al.. New insights into the pathogenesis and nonsurgical management of Graves orbitopathy. Nat Rev Endocrinol. 2020; 16: 104–116. - PubMed
MeSH terms
LinkOut - more resources
Full Text Sources
Miscellaneous

