A bioinspired and degradable riboflavin-containing polypeptide as a sustainable material for energy storage
- PMID: 40549907
- PMCID: PMC12232413
- DOI: 10.1073/pnas.2509325122
A bioinspired and degradable riboflavin-containing polypeptide as a sustainable material for energy storage
Abstract
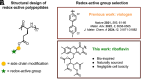
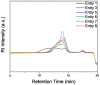
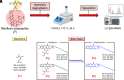
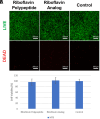
Inspired by Nature, we present a polypeptide-based organic redox-active material constructed from renewable feedstocks, L-glutamic acid (an amino acid) and riboflavin (vitamin B2), to address challenges with start-to-end-of-life management in energy storage systems (ESSs). The amino acid was utilized to establish a degradable polymer backbone, along which many copies of riboflavin were incorporated to serve as the redox-active pendant groups that enabled energy storage. The overall synthesis involved the ring-opening polymerization (ROP) of an l-glutamic acid-derived N-carboxyanhydride (NCA) monomer, followed by side chain activation with azides and, finally, click coupling to achieve installation of alkyne-functionalized riboflavin moieties. The steric bulkiness and rich chemical functionality of riboflavin resulted in synthetic complexities that required reaction optimization to achieve the desired polymer structure. Electrochemical characterization of the resultant riboflavin polypeptide, in organic electrolyte, showed quasireversible redox activity with a half-wave potential (E1/2) of ca. -1.10 V vs. ferrocene/ferrocenium (Fc/Fc+). Cell viability assays revealed biocompatibility, as indicated by negligible cytotoxicity for fibroblast cells. The polypeptide design, consisting of labile amide backbone linkages and side-chain ester functionalities that tethered the riboflavin units to the backbone, enabled hydrolytic degradation to recover building blocks for future upcycling or recycling. This bioinspired strategy advances the development of degradable redox-active polymers and promotes sustainable materials design for circular energy storage technologies.
Keywords: bioderived redox-active material; controlled ring-opening polymerization; degradable polymers; sustainable energy storage.
Conflict of interest statement
Competing interests statement:The authors declare no competing interest.
Figures



References
-
- Wen J., Yu Y., Chen C., A review on lithium-ion batteries safety issues: Existing problems and possible solutions. Mater. Express 2, 197–212 (2012).
-
- Mrozik W., Rajaeifar M. A., Heidrich O., Christensen P., Environmental impacts, pollution sources and pathways of spent lithium-ion batteries. Energy Environ. Sci. 14, 6099–6121 (2021).
-
- Olivetti E. A., Ceder G., Gaustad G. G., Fu X., Lithium-ion battery supply chain considerations: Analysis of potential bottlenecks in critical metals. Joule 1, 229–243 (2017).
-
- Ma T., et al. , Nonconjugated redox-active polymers: Electron transfer mechanisms, energy storage, and chemical versatility. Annu. Rev. Chem. Biomol. Eng. 14, 187–216 (2023). - PubMed
-
- Kim J., Kim J. H., Ariga K., Redox-active polymers for energy storage nanoarchitectonics. Joule 1, 739–768 (2017).
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

