Lipid metabolism in the adrenal gland
- PMID: 40551885
- PMCID: PMC12183020
- DOI: 10.3389/fendo.2025.1577505
Lipid metabolism in the adrenal gland
Abstract
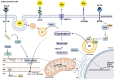
The adrenal gland consists of the medulla and the cortex. The chromaffin cells of the adrenal medulla release catecholamines via regulated exocytosis. Vesicle formation, trafficking, maturation and fusion with the plasma membrane are orchestrated by lipids such as cholesterol, diacylglycerol, phosphatidic acid and phosphatidylinositol-4,5-bisphosphate. On the other hand, the adrenal cortex is a highly specialized lipid-metabolizing organ secreting steroid hormones. Cholesterol, acquired from circulating lipoproteins and de novo biosynthesis, is mobilized from intracellular stores and transported to mitochondria to be used as a substrate for steroidogenesis. Steroidogenesis is regulated by free polyunsaturated fatty acids (PUFA) and an increased PUFA content in phospholipids promotes steroidogenesis. Cholesterol efflux and lipid-processing macrophages further contribute to lipid homeostasis in the adrenal gland. Given that lipidomics have revolutionized our perception of cell function, we anticipate that this will also hold true for the investigation of adrenocortical function. Such investigations may pinpoint novel targets for the management of abnormal adrenal function.
Keywords: adrenal cortex; adrenal medulla; aldosterone; cholesterol metabolism; cortisol; lipid metabolism; phospholipids.
Copyright © 2025 Aderhold and Alexaki.
Conflict of interest statement
The authors declare absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
Similar articles
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
Interventions for infantile haemangiomas of the skin.Cochrane Database Syst Rev. 2018 Apr 18;4(4):CD006545. doi: 10.1002/14651858.CD006545.pub3. Cochrane Database Syst Rev. 2018. PMID: 29667726 Free PMC article.
-
Comparison of cellulose, modified cellulose and synthetic membranes in the haemodialysis of patients with end-stage renal disease.Cochrane Database Syst Rev. 2001;(3):CD003234. doi: 10.1002/14651858.CD003234. Cochrane Database Syst Rev. 2001. Update in: Cochrane Database Syst Rev. 2005 Jul 20;(3):CD003234. doi: 10.1002/14651858.CD003234.pub2. PMID: 11687058 Updated.
-
Adenovirus Modulates Toll-Like Receptor 4 Signaling by Reprogramming ORP1L-VAP Protein Contacts for Cholesterol Transport from Endosomes to the Endoplasmic Reticulum.J Virol. 2017 Feb 28;91(6):e01904-16. doi: 10.1128/JVI.01904-16. Print 2017 Mar 15. J Virol. 2017. PMID: 28077646 Free PMC article.
-
Digital interventions to improve adherence to maintenance medication in asthma.Cochrane Database Syst Rev. 2022 Jun 13;6(6):CD013030. doi: 10.1002/14651858.CD013030.pub2. Cochrane Database Syst Rev. 2022. PMID: 35691614 Free PMC article.
References
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources

