Machine learning-based radiomics models for the prediction of metachronous liver metastases in patients with colorectal cancer: A multimodal study
- PMID: 40552255
- PMCID: PMC12183777
- DOI: 10.3892/ol.2025.15140
Machine learning-based radiomics models for the prediction of metachronous liver metastases in patients with colorectal cancer: A multimodal study
Abstract
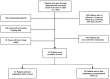
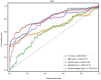
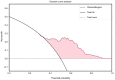
The aim of the present study was to investigate whether a multimodal radiomics model powered by machine learning could accurately predict the occurrence of metachronous liver metastasis (MLM) in patients with colorectal cancer (CRC). A total of 157 patients diagnosed with CRC between 2010 and 2020 were retrospectively included in the present study; of these patients, 67 patients developed liver metastases within 2 years of treatment, while the remaining patients (n=90) did not. Radiomics features were extracted from annotated MR images of the tumor and portal venous phase CT images of the liver in each patient. Subsequently, machine learning-based radiomics models were developed and integrated with the clinical features for MLM prediction, employing Least Absolute Shrinkage and Selection Operator and Random Forest algorithms. The performance of the models were evaluated using the receiver operating characteristic curve analysis, while the clinical utility was measured using the decision curve analysis. A total of 922 and 1,082 radiomics features were extracted from the MR and CT images of each patient, respectively, which quantified the intensity, shape, orientation and texture of the tumor and liver. The mean area under the curve (AUC) values for the prediction of MLM were 0.80, 0.68 and 0.82 for the CT, MRI and merged models, respectively. For the clinical and clinical-merged models, the AUC values were 0.62 and 0.75, respectively. There was no significant difference between the CT model and the merged model (P>0.05). In conclusion, the preliminary results of the present study demonstrated the utility of machine learning-based radiomics models in the prediction of MLM in patients with CRC. However, further research is warranted to explore the potential of multimodal fusion models, due to the minimal improvement observed in diagnostic performance.
Keywords: colorectal cancer; machine learning; metachronous liver metastasis; multimodal; radiomics.
Copyright: © 2025 Wang et al.
Conflict of interest statement
The authors declare that they have no competing interests.
Figures




Similar articles
-
Machine learning-based CT radiomics enhances bladder cancer staging predictions: A comparative study of clinical, radiomics, and combined models.Med Phys. 2024 Sep;51(9):5965-5977. doi: 10.1002/mp.17288. Epub 2024 Jul 8. Med Phys. 2024. PMID: 38977273
-
Prediction of KRAS gene mutations in colorectal cancer using a CT-based radiomic model.Front Med (Lausanne). 2025 May 12;12:1592497. doi: 10.3389/fmed.2025.1592497. eCollection 2025. Front Med (Lausanne). 2025. PMID: 40421293 Free PMC article.
-
CT-Based radiomics and deep learning for the preoperative prediction of peritoneal metastasis in ovarian cancers.Abdom Radiol (NY). 2025 Aug 13. doi: 10.1007/s00261-025-05162-w. Online ahead of print. Abdom Radiol (NY). 2025. PMID: 40802053
-
Magnetic resonance perfusion for differentiating low-grade from high-grade gliomas at first presentation.Cochrane Database Syst Rev. 2018 Jan 22;1(1):CD011551. doi: 10.1002/14651858.CD011551.pub2. Cochrane Database Syst Rev. 2018. PMID: 29357120 Free PMC article.
-
Eliciting adverse effects data from participants in clinical trials.Cochrane Database Syst Rev. 2018 Jan 16;1(1):MR000039. doi: 10.1002/14651858.MR000039.pub2. Cochrane Database Syst Rev. 2018. PMID: 29372930 Free PMC article.
References
-
- Horak J, Kubecek O, Siskova A, Honkova K, Chvojkova I, Krupova M, Manethova M, Vodenkova S, García-Mulero S, John S, et al. Differences in genome, transcriptome, miRNAome, and methylome in synchronous and metachronous liver metastasis of colorectal cancer. Front Oncol. 2023;13:1133598. doi: 10.3389/fonc.2023.1133598. - DOI - PMC - PubMed
-
- Xu N, Gao Z, Wu D, Chen H, Zhang Z, Zhang L, Wang Y, Lu X, Yao X, Liu X, et al. 5-hydroxymethylcytosine features of portal venous blood predict metachronous liver metastases of colorectal cancer and reveal phosphodiesterase 4 as a therapeutic target. Clin Transl Med. 2025;15:e70189. doi: 10.1002/ctm2.70189. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
