TRPV4 activation by TGFβ2 enhances cellular contractility and drives ocular hypertension
- PMID: 40552711
- PMCID: PMC12187138
- DOI: 10.7554/eLife.104894
TRPV4 activation by TGFβ2 enhances cellular contractility and drives ocular hypertension
Abstract
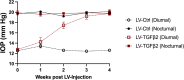
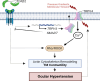
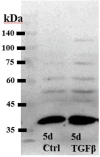
The risk for developing primary open-angle glaucoma (POAG) correlates with the magnitude of ocular hypertension (OHT) and the concentration of transforming growth factor-β2 (TGFβ2) in the aqueous humor. Effective treatment of POAG requires a detailed understanding of the interaction between pressure sensing mechanisms in the trabecular meshwork (TM) and biochemical risk factors. Here, we employed molecular, optical, electrophysiological, and tonometric strategies to establish the role of TGFβ2 in transcription and functional expression of mechanosensitive channel isoforms alongside studies of TM contractility in biomimetic hydrogels and intraocular pressure (IOP) regulation in a mouse model of TGFβ2-induced OHT. TGFβ2 upregulated expression of Trpv4 and Piezo1 transcripts and time-dependently augmented functional TRPV4 activation. TRPV4 agonists induced contractility of TM-seeded hydrogels, whereas pharmacological inhibition suppressed TGFβ2-induced hypercontractility and abrogated OHT in eyes overexpressing TGFβ2. Trpv4-deficient mice resisted TGFβ2-driven increases in IOP, but nocturnal OHT was not additive to TGFβ-evoked OHT. Our study establishes the fundamental role of TGFβ as a modulator of mechanosensing in nonexcitable cells, identifies the TRPV4 channel as the final common mechanism for TM contractility and circadian and pathological OHT, and offers insights for future treatments that can lower IOP in the sizeable cohort of hypertensive glaucoma patients that resist current treatments.
Keywords: TGFB2; TRPV4; calcium; circadian rhythm; contractility; electrophysiology; glaucoma; human (cells); intraocular pressure; knockout mice; mouse; neuroscience.
© 2025, Rudzitis et al.
Conflict of interest statement
CR, ML, AS, SR, DK, YT, MD, WS, SH No competing interests declared, DK Co-founder of TMClear and co-inventor of patents (US 2015/0133411, US20230026696) related to the development of cornea permeant TRPV4 channel antagonists; the patents were licensed to TMClear by the University of Utah
Figures



Update of
-
TRPV4 overactivation enhances cellular contractility and drives ocular hypertension in TGFβ2 overexpressing eyes.bioRxiv [Preprint]. 2025 May 19:2024.11.05.622187. doi: 10.1101/2024.11.05.622187. bioRxiv. 2025. Update in: Elife. 2025 Jun 24;14:RP104894. doi: 10.7554/eLife.104894. PMID: 39574569 Free PMC article. Updated. Preprint.
Similar articles
-
TRPV4 controls circadian and pathological ocular hypertension.J Physiol. 2025 Jul;603(14):4091-4111. doi: 10.1113/JP288706. Epub 2025 Jul 10. J Physiol. 2025. PMID: 40638660 Free PMC article.
-
TRPV4 overactivation enhances cellular contractility and drives ocular hypertension in TGFβ2 overexpressing eyes.bioRxiv [Preprint]. 2025 May 19:2024.11.05.622187. doi: 10.1101/2024.11.05.622187. bioRxiv. 2025. Update in: Elife. 2025 Jun 24;14:RP104894. doi: 10.7554/eLife.104894. PMID: 39574569 Free PMC article. Updated. Preprint.
-
TRPV4 and chloride channels mediate volume sensing in trabecular meshwork cells.Am J Physiol Cell Physiol. 2024 Aug 1;327(2):C403-C414. doi: 10.1152/ajpcell.00295.2024. Epub 2024 Jun 17. Am J Physiol Cell Physiol. 2024. PMID: 38881423 Free PMC article.
-
Rho kinase inhibitor for primary open-angle glaucoma and ocular hypertension.Cochrane Database Syst Rev. 2022 Jun 10;6(6):CD013817. doi: 10.1002/14651858.CD013817.pub2. Cochrane Database Syst Rev. 2022. PMID: 35686679 Free PMC article.
-
Role of Glucocorticoids and Glucocorticoid Receptors in Glaucoma Pathogenesis.Cells. 2023 Oct 14;12(20):2452. doi: 10.3390/cells12202452. Cells. 2023. PMID: 37887296 Free PMC article. Review.
Cited by
-
TRPV4 controls circadian and pathological ocular hypertension.J Physiol. 2025 Jul;603(14):4091-4111. doi: 10.1113/JP288706. Epub 2025 Jul 10. J Physiol. 2025. PMID: 40638660 Free PMC article.
References
-
- Adapala RK, Thoppil RJ, Luther DJ, Paruchuri S, Meszaros JG, Chilian WM, Thodeti CK. TRPV4 channels mediate cardiac fibroblast differentiation by integrating mechanical and soluble signals. Journal of Molecular and Cellular Cardiology. 2013;54:45–52. doi: 10.1016/j.yjmcc.2012.10.016. - DOI - PMC - PubMed
-
- Alessandri-Haber N, Dina OA, Joseph EK, Reichling D, Levine JD. A transient receptor potential vanilloid 4-dependent mechanism of hyperalgesia is engaged by concerted action of inflammatory mediators. The Journal of Neuroscience. 2006;26:3864–3874. doi: 10.1523/JNEUROSCI.5385-05.2006. - DOI - PMC - PubMed
MeSH terms
Substances
Grants and funding
- R01EY005722/EY/NEI NIH HHS/United States
- T32EY024234/EY/NEI NIH HHS/United States
- R21EY036189/NH/NIH HHS/United States
- P30 EY005722/EY/NEI NIH HHS/United States
- R01 EY022076/EY/NEI NIH HHS/United States
- R01EY022076/EY/NEI NIH HHS/United States
- R01 EY022359/EY/NEI NIH HHS/United States
- R01EY022359/EY/NEI NIH HHS/United States
- R21 EY036189/EY/NEI NIH HHS/United States
- T32 EY024234/EY/NEI NIH HHS/United States
- R0EY1031817/EY/NEI NIH HHS/United States
- R01EY034096/EY/NEI NIH HHS/United States
- P30 EY014800/EY/NEI NIH HHS/United States
- R01 EY034096/EY/NEI NIH HHS/United States
- P30EY014800/EY/NEI NIH HHS/United States
LinkOut - more resources
Full Text Sources

