S1PR1 regulates lymphatic valve development and tertiary lymphoid organ formation in the ileum
- PMID: 40553105
- PMCID: PMC12187108
- DOI: 10.1084/jem.20241799
S1PR1 regulates lymphatic valve development and tertiary lymphoid organ formation in the ileum
Abstract
Efficient lymph flow is ensured by lymphatic valves (LVs). The mechanisms that regulate LV development are incompletely understood. Here, we show that the deletion of the GPCR sphingosine 1-phosphate receptor-1 (S1PR1) from lymphatic endothelial cells (LECs) results in fewer LVs. Interestingly, LVs that remained in the terminal ileum-draining lymphatic vessels were specifically dysfunctional. Furthermore, tertiary lymphoid organs (TLOs) formed in the terminal ileum of the mutant mice. TLOs in this location are associated with ileitis in humans and mice. However, mice lacking S1PR1 did not develop obvious characteristics of ileitis. Mechanistically, S1PR1 regulates shear stress signaling and the expression of the valve-regulatory molecules FOXC2 and connexin-37. Importantly, Foxc2+/- mice, a model for lymphedema-distichiasis syndrome, also develop TLOs in the terminal ileum. Thus, we have discovered S1PR1 as a previously unknown regulator of LV and TLO development. We also suggest that TLOs are a sign of subclinical inflammation that can form due to lymphatic disorders in the absence of ileitis.
© 2025 Geng et al.
Conflict of interest statement
Disclosures: The authors declare no competing interests exist.
Figures


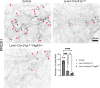





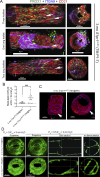







References
-
- Bell, R., Brice G., Child A.H., Murday V.A., Mansour S., Sandy C.J., Collin J.R., Brady A.F., Callen D.F., Burnand K., et al. 2001. Analysis of lymphoedema-distichiasis families for FOXC2 mutations reveals small insertions and deletions throughout the gene. Hum. Genet. 108:546–551. 10.1007/s004390100528 - DOI - PubMed
MeSH terms
Substances
Grants and funding
- 4340/Tobacco Settlement Endowment Trust
- R01 HL133216/HL/NHLBI NIH HHS/United States
- R01HL131652/National Heart, Lung and Blood Institute
- Oklahoma Center for Adult Stem Cell Research
- R01HL153728/National Heart, Lung and Blood Institute
- R01HL163095/National Heart, Lung and Blood Institute
- ANR-23-CE14-0035-2/French National Research Agency
- P30 GM149376/GM/NIGMS NIH HHS/United States
- R01 HL131652/HL/NHLBI NIH HHS/United States
- R01HL175083/National Heart, Lung and Blood Institute
- R01HL122578/National Heart, Lung and Blood Institute
- R01 HL163095/HL/NHLBI NIH HHS/United States
- R01 HL122578/HL/NHLBI NIH HHS/United States
- NH/NIH HHS/United States
- R01 HL153728/HL/NHLBI NIH HHS/United States
- P20 GM139763/GM/NIGMS NIH HHS/United States
- R01 HL175083/HL/NHLBI NIH HHS/United States
- R01HL133216/National Heart, Lung and Blood Institute
- ANR-19-CE14-0028-01/French National Research Agency
LinkOut - more resources
Full Text Sources
Other Literature Sources
Molecular Biology Databases
Research Materials

