Experimental research on fungal inhibition using dissolving microneedles of terbinafine hydrochloride nanoemulsion for beta-1,3-glucanase
- PMID: 40556857
- PMCID: PMC12183721
- DOI: 10.1039/d5na00163c
Experimental research on fungal inhibition using dissolving microneedles of terbinafine hydrochloride nanoemulsion for beta-1,3-glucanase
Abstract
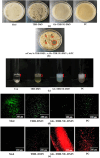
Objective: Onychomycosis is characterized by high transmission and low recovery rates, leading to a lack of optimal treatments. This study used dissolving microneedles as drug carriers to design dual-loaded DMN patches with β-1,3-glucanase and terbinafine hydrochloride nanoemulsion. Methods: Dissolving microneedles of terbinafine hydrochloride nanoemulsion for beta-1,3-glucanase (Gls-TBH-NE-DMN) were prepared using the centrifugal molding method. Parafilm®, weight method, mouse skin puncture experiments, and mouse skin tissue sections were used to evaluate the mechanical properties of Gls-TBH-NE-DMN. Gls-TBH-NE-DMN was tested for its in vitro anti-Candida albicans susceptibility, ability to inhibit fungal cell wall synthesis, and fungal biofilm penetration and inhibition capabilities. The in vivo inhibitory effect of Gls-TBH-NE-DMN on fungi was studied using a rabbit nail infection model. Results: Gls-TBH-NE-DMN exhibited a penetration rate of 98% on skin simulants and a compressive bending resistance of 12.75%. It was nonirritating to the dorsal skin of mice, with no edema or erythema on the surface of the skin, suggesting a good safety profile. The accumulative release of Gls-TBH-NE-DMN at 72 h was 78.74% ± 0.64%, and that of terbinafine hydrochloride dissolving microneedles was 49.52% ± 0.80%. In the in vitro transdermal permeation experiment, the cumulative transdermal permeation of Gls-TBH-NE-DMN at 72 h was 73.21% ± 0.84% and that of terbinafine hydrochloride (TBH) was 20.57% ± 0.98%. In vitro inhibition of C. albicans showed that the lowest inhibitory concentration in the Gls-TBH-NE-DMN group was 512 μg mL-1. Furthermore, experiments showed that Gls-TBH-NE-DMN could effectively inhibit fungal cell wall synthesis and disrupt the C. albicans biofilm, inhibiting fungal growth. Conclusions: Gls-TBH-NE-DMN prepared in this study provides new ideas for treating skin fungal diseases and for developing antimicrobial drug formulations.
This journal is © The Royal Society of Chemistry.
Conflict of interest statement
The authors have no relevant financial or non-financial interests to disclose.
Figures




References
LinkOut - more resources
Full Text Sources
Miscellaneous

