Evaluation of the Potential of Metal-Organic Compounds ZIF-8 and F300 in a Membrane Filtration-Adsorption Process for the Removal of Antibiotics from Water
- PMID: 40558209
- PMCID: PMC12189444
- DOI: 10.3390/antibiotics14060619
Evaluation of the Potential of Metal-Organic Compounds ZIF-8 and F300 in a Membrane Filtration-Adsorption Process for the Removal of Antibiotics from Water
Abstract
Background/objectives: Antibiotic contamination in water sources is a growing global concern, contributing to environmental degradation and the proliferation of antimicrobial resistance. Traditional treatment methods, such as advanced oxidation or high-pressure membrane processes, are often energy-intensive and economically unsustainable for large-scale or decentralized applications. This study explores the potential of two cost-effective, commercially available metal-organic frameworks (MOFs), ZIF-8 and F300, to improve the performance of membrane-based filtration-adsorption systems for removing tetracycline and sulfadiazine from water.
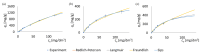
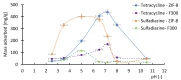
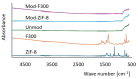
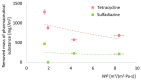
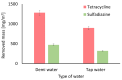
Methods: Batch adsorption experiments were performed to evaluate the uptake capacities, kinetics, and isotherms of both MOFs toward the selected antibiotics. The membranes were modified using a low-cost silane-assisted deposition of MOF particles and tested in a microfiltration system. Removal efficiencies and water permeability were assessed and kinetic and isotherm models were applied to understand the adsorption mechanisms.
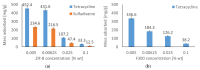
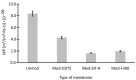
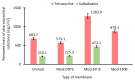
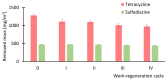
Results: ZIF-8 showed superior adsorption performance, with maximum capacities of 442.2 mg/g for tetracycline and 219.3 mg/g for sulfadiazine. F300 was effective only for tetracycline. Membranes modified with ZIF-8 improved pharmaceutical removal by 187% (tetracycline) and 224% (sulfadiazine) compared to unmodified membranes. Although permeability decreased due to increased hydrophobicity, the materials and processes remained economically favorable.
Conclusions: This study demonstrates that MOF-modified ceramic membranes, particularly those incorporating ZIF-8, offer a low-cost, scalable, and energy-efficient alternative for pharmaceutical removal from water. The approach combines strong environmental impact with economic viability, making it attractive for broader implementation in water treatment systems.
Keywords: MOF; adsorption; filtration; membranes; sulfadiazine; tetracycline; water.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
Comparison of cellulose, modified cellulose and synthetic membranes in the haemodialysis of patients with end-stage renal disease.Cochrane Database Syst Rev. 2001;(3):CD003234. doi: 10.1002/14651858.CD003234. Cochrane Database Syst Rev. 2001. Update in: Cochrane Database Syst Rev. 2005 Jul 20;(3):CD003234. doi: 10.1002/14651858.CD003234.pub2. PMID: 11687058 Updated.
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of topotecan for ovarian cancer.Health Technol Assess. 2001;5(28):1-110. doi: 10.3310/hta5280. Health Technol Assess. 2001. PMID: 11701100
-
Graphene oxide/chitosan hydrogels for removal of antibiotics.Environ Technol. 2025 Jul;46(17):3391-3421. doi: 10.1080/09593330.2025.2464267. Epub 2025 Feb 22. Environ Technol. 2025. PMID: 39985820 Review.
-
Cost-effectiveness of using prognostic information to select women with breast cancer for adjuvant systemic therapy.Health Technol Assess. 2006 Sep;10(34):iii-iv, ix-xi, 1-204. doi: 10.3310/hta10340. Health Technol Assess. 2006. PMID: 16959170
-
Intravenous magnesium sulphate and sotalol for prevention of atrial fibrillation after coronary artery bypass surgery: a systematic review and economic evaluation.Health Technol Assess. 2008 Jun;12(28):iii-iv, ix-95. doi: 10.3310/hta12280. Health Technol Assess. 2008. PMID: 18547499
References
-
- Chaturvedi P., Shukla P., Giri B.S., Chowdhary P., Chandra R., Gupta P., Pandey A. Prevalence and hazardous impact of pharmaceutical and personal care products and antibiotics in environment: A review on emerging contaminants. Environ. Res. 2021;194:110664. doi: 10.1016/j.envres.2020.110664. - DOI - PubMed
-
- Serwecińska L. Antimicrobials and Antibiotic-Resistant Bacteria: A Risk to the Environment and to Public Health. Water. 2020;12:3313. doi: 10.3390/w12123313. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources

