Selective Blockade of Two Aquaporin Channels, AQP3 and AQP9, Impairs Human Leukocyte Migration
- PMID: 40558507
- PMCID: PMC12191162
- DOI: 10.3390/cells14120880
Selective Blockade of Two Aquaporin Channels, AQP3 and AQP9, Impairs Human Leukocyte Migration
Abstract
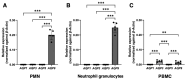
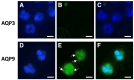
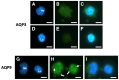
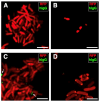
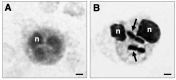
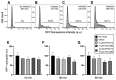
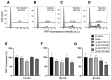
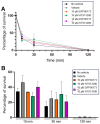
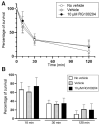
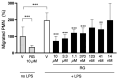
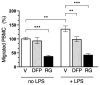
Peripheral blood leukocytes are able to migrate to the inflamed tissue, and to engulf and kill invading microbes. This requires rapid modifications of cell morphology and volume through fast movements of osmotic water into or out of the cell. In this process, membrane water channels, aquaporins (AQPs), are critical for cell shape changes as AQP-mediated water movement indirectly affects the cell cytoskeleton and, thereby, the signaling cascades. Recent studies have shown that the deletion or gating of two immune cell AQPs, AQP3 and AQP9, impairs inflammation and improves survival in microbial sepsis. Here, we assessed the expression and distribution of AQP3 and AQP9 in human leukocytes and investigated their involvement in the phagocytosis and killing of the Gram-negative pathogenic bacterium Klebsiella pneumoniae, and their role in lipopolysaccharide (LPS)-induced cell migration. By RT-qPCR, AQP3 mRNA was found in peripheral blood mononuclear cells (PBMCs) but it was undetectable in polymorphonuclear white blood cells (PMNs). AQP9 was found both in PBMCs and PMNs, particularly in neutrophil granulocytes. Immunofluorescence confirmed the AQP3 expression in monocytes and, to a lesser degree, in lymphocytes. AQP9 was expressed both in PBMCs and neutrophils. Specific inhibitors of AQP3 (DFP00173) and AQP9 (HTS13286 and RG100204) were used for bacterial phagocytosis and killing studies. No apparent involvement of individually blocked AQP3 or AQP9 was observed in the phagocytosis of K. pneumoniae by neutrophils or monocytes after 10, 30, or 60 min of bacterial infection. A significant impairment in the phagocytic capacity of monocytes but not neutrophils was observed only when both AQPs were inhibited simultaneously and when the infection lasted for 60 min. No impairment in bacterial clearance was found when AQP3 and AQP9 were individually or simultaneously blocked. PBMC migration was significantly impaired after exposure to the AQP9 blocker RG100204 in the presence or absence of LPS. The AQP3 inhibitor DFP00173 reduced PBMC migration only under LPS exposure. Neutrophil migration was considerably reduced in the presence of RG100204 regardless of whether there was an LPS challenge or not. Taken together, these results indicate critical but distinct involvements for AQP3 and AQP9 in leukocyte motility, while no roles are played in bacterial killing. Further studies are needed in order to understand the precise ways in which these two AQPs intervene during bacterial infections.
Keywords: LPS; aquaporin inhibitors; cell motility; host–bacteria relationship; inflammation; innate immunity; phagocytosis; white blood cells.
Conflict of interest statement
Author Michael Rützler was employed by the company Apoglyx AB. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Comparison of cellulose, modified cellulose and synthetic membranes in the haemodialysis of patients with end-stage renal disease.Cochrane Database Syst Rev. 2001;(3):CD003234. doi: 10.1002/14651858.CD003234. Cochrane Database Syst Rev. 2001. Update in: Cochrane Database Syst Rev. 2005 Jul 20;(3):CD003234. doi: 10.1002/14651858.CD003234.pub2. PMID: 11687058 Updated.
-
Intravenous magnesium sulphate and sotalol for prevention of atrial fibrillation after coronary artery bypass surgery: a systematic review and economic evaluation.Health Technol Assess. 2008 Jun;12(28):iii-iv, ix-95. doi: 10.3310/hta12280. Health Technol Assess. 2008. PMID: 18547499
-
Home treatment for mental health problems: a systematic review.Health Technol Assess. 2001;5(15):1-139. doi: 10.3310/hta5150. Health Technol Assess. 2001. PMID: 11532236
-
Antiretrovirals for reducing the risk of mother-to-child transmission of HIV infection.Cochrane Database Syst Rev. 2011 Jul 6;(7):CD003510. doi: 10.1002/14651858.CD003510.pub3. Cochrane Database Syst Rev. 2011. PMID: 21735394
-
Antiretrovirals for reducing the risk of mother-to-child transmission of HIV infection.Cochrane Database Syst Rev. 2007 Jan 24;(1):CD003510. doi: 10.1002/14651858.CD003510.pub2. Cochrane Database Syst Rev. 2007. Update in: Cochrane Database Syst Rev. 2011 Jul 06;(7):CD003510. doi: 10.1002/14651858.CD003510.pub3. PMID: 17253490 Updated.
Cited by
-
High-Calorie Diets Exacerbate Lipopolysaccharide-Induced Pneumonia by Promoting Propionate-Mediated Neutrophil Extracellular Traps.Nutrients. 2025 Jul 7;17(13):2242. doi: 10.3390/nu17132242. Nutrients. 2025. PMID: 40647346 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

