Mitogen-Activated Protein Kinase Kinase Kinase 1 Overexpression Disrupts Development of the Ocular Surface Epithelium
- PMID: 40558521
- PMCID: PMC12190364
- DOI: 10.3390/cells14120894
Mitogen-Activated Protein Kinase Kinase Kinase 1 Overexpression Disrupts Development of the Ocular Surface Epithelium
Abstract
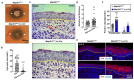
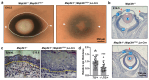
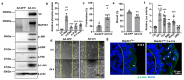
Mitogen-Activated Protein Kinase Kinase Kinase 1 (MAP3K1) is a key signaling molecule essential for eyelid closure during embryogenesis. In mice, Map3k1 knockout leads to a fully penetrant eye-open at birth (EOB) phenotype due to disrupted MAPK signaling, abnormal epithelial differentiation, and morphogenesis. To further explore the roles of MAP3K1 in ocular development, we generated a Cre-inducible gain-of-function transgenic mouse, designated as Map3k1TG, and crossed it with Lens epithelial (Le)-Cre mice to drive MAP3K1 overexpression in developing ocular surface epithelium (OSE). Map3k1TG;Le-Cre embryos exhibited ocular defects including premature eyelid closure, lens degeneration, and corneal edema. While corneal epithelial differentiation remained intact, the lens epithelium degenerated with lens formation compromised. Eyelid epithelium was markedly thickened, containing cells with aberrant keratin (K)14/K10 co-expression. Genetic rescue experiments revealed that Map3k1TG;Le-Cre restored eyelid closure in Map3k1 knockout mice, whereas MAP3K1 deficiency attenuated the epithelial thickening caused by transgene expression. Mechanistically, MAP3K1 overexpression enhanced c-Jun phosphorylation in vivo and activated JNK-c-Jun, WNT, TGFβ, and Notch signaling and promoted keratinocyte proliferation and migration in vitro. These findings highlight a dose-sensitive role for MAP3K1 in regulating epithelial proliferation, differentiation, and morphogenesis during eyelid development.
Keywords: MAP3K1 gain-of-function; epithelial morphogenesis; eyelid closure; ocular surface epithelium.
Conflict of interest statement
The authors declare that no competing interests exist.
Figures



Similar articles
-
Mitogen-activated protein kinase kinase kinase 1 (MAP3K1) integrates developmental signals for eyelid closure.Proc Natl Acad Sci U S A. 2011 Oct 18;108(42):17349-54. doi: 10.1073/pnas.1102297108. Epub 2011 Oct 3. Proc Natl Acad Sci U S A. 2011. PMID: 21969564 Free PMC article.
-
Epithelial sheet movement requires the cooperation of c-Jun and MAP3K1.Dev Biol. 2014 Nov 1;395(1):29-37. doi: 10.1016/j.ydbio.2014.09.001. Epub 2014 Sep 16. Dev Biol. 2014. PMID: 25224220 Free PMC article.
-
Gene-Environment Interactions Target Mitogen-activated Protein 3 Kinase 1 (MAP3K1) Signaling in Eyelid Morphogenesis.J Biol Chem. 2015 Aug 7;290(32):19770-9. doi: 10.1074/jbc.M115.665729. Epub 2015 Jun 24. J Biol Chem. 2015. PMID: 26109068 Free PMC article.
-
Shroom3 facilitates optic fissure closure via tissue alignment and reestablishment of apical-basal polarity during epithelial fusion.Dev Biol. 2025 Jun;522:91-105. doi: 10.1016/j.ydbio.2025.03.008. Epub 2025 Mar 18. Dev Biol. 2025. PMID: 40113025 Free PMC article. Review.
-
Amniotic membrane transplantation for acute ocular burns.Cochrane Database Syst Rev. 2022 Sep 1;9(9):CD009379. doi: 10.1002/14651858.CD009379.pub3. Cochrane Database Syst Rev. 2022. PMID: 36047788 Free PMC article.
References
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

