Quantitative Ultrashort Echo Time Magnetization Transfer Imaging of the Osteochondral Junction: An In Vivo Knee Osteoarthritis Study
- PMID: 40558797
- PMCID: PMC12193826
- DOI: 10.3390/jimaging11060198
Quantitative Ultrashort Echo Time Magnetization Transfer Imaging of the Osteochondral Junction: An In Vivo Knee Osteoarthritis Study
Abstract
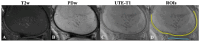
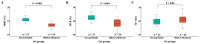
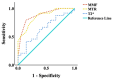
Osteoarthritis (OA) is the most prevalent degenerative joint disorder worldwide, causing significant declines in quality of life. The osteochondral junction (OCJ), a critical structural interface between deep cartilage and subchondral bone, plays an essential role in OA progression but is challenging to assess using conventional magnetic resonance imaging (MRI) due to its short T2 relaxation times. This study aimed to evaluate the utility of ultrashort echo time (UTE) MRI biomarkers, including macromolecular fraction (MMF), magnetization transfer ratio (MTR), and T2*, for in vivo quantification of OCJ changes in knee OA for the first time. Forty-five patients (mean age: 53.8 ± 17.0 years, 50% female) were imaged using 3D UTE-MRI sequences on a 3T clinical MRI scanner. Patients were stratified into two OA groups based on radiographic Kellgren-Lawrence (KL) scores: normal/subtle (KL = 0-1) (n = 21) and mild to moderate (KL = 2-3) (n = 24). Quantitative analysis revealed significantly lower MMF (15.8 ± 1.4% vs. 13.6 ± 1.2%, p < 0.001) and MTR (42.5 ± 2.5% vs. 38.2 ± 2.3%, p < 0.001) in the higher KL 2-3 group, alongside a higher trend in T2* values (19.7 ± 2.6 ms vs. 21.6 ± 3.8 ms, p = 0.06). Moreover, MMF and MTR were significantly negatively correlated with KL grades (r = -0.66 and -0.59; p < 0.001, respectively), while T2* showed a weaker positive correlation (r = 0.26, p = 0.08). Receiver operating characteristic (ROC) analysis demonstrated superior diagnostic accuracy for MMF (AUC = 0.88) and MTR (AUC = 0.86) compared to T2* (AUC = 0.64). These findings highlight UTE-MT techniques (i.e., MMF and MTR) as promising imaging tools for detecting OCJ degeneration in knee OA, with potential implications for earlier and more accurate diagnosis and disease monitoring.
Keywords: macromolecular fraction (MMF); magnetization transfer ratio (MTR); osteoarthritis (OA); osteochondral junction (OCJ); ultrashort echo time (UTE) MRI.
Conflict of interest statement
The authors declare no conflict of interest.
Figures

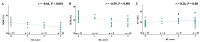
References
Grants and funding
LinkOut - more resources
Full Text Sources

