Effects of a Proteinase Inhibitor from Inga laurina Seeds (ILTI) on Aedes aegypti Larval Development
- PMID: 40558860
- PMCID: PMC12194339
- DOI: 10.3390/jox15030077
Effects of a Proteinase Inhibitor from Inga laurina Seeds (ILTI) on Aedes aegypti Larval Development
Abstract
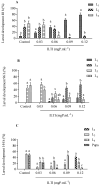
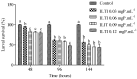
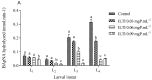
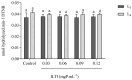
Aedes aegypti (Linnaeus, 1762) is Brazil's primary vector of epidemiologically significant arboviruses such as yellow fever, dengue, Zika, and chikungunya. Despite using conventional chemical control measures, this species has developed resistance to standard chemical insecticides, prompting the search for natural larvicidal compounds. Plant protease inhibitors offer an insecticidal alternative as the primary digestive enzymes in the midgut of Ae. aegypti are proteases (trypsin and chymotrypsin). Ae. aegypti larvae fed with ILTI, a Kunitz-type trypsin inhibitor derived from Inga laurina seeds, at concentrations between 0.03 mg of protein per mL (mgP/mL) and 0.12 mgP/mL, exhibited delayed larval development, with a lethal concentration (LC50) of 0.095 mgP mL-1 of ILTI for 50% of fourth-instar larvae (L4). The ex vivo assay indicated that ILTI effectively inhibited the activity of larval trypsin, which remained susceptible to the inhibitor. Additionally, molecular modelling and docking studies were conducted to predict the three-dimensional ILTI/enzyme molecular complexes at the atomic level. Therefore, the results demonstrate that ILTI functions as a protease inhibitor in this species, presenting itself as a promising larvicidal tool in the control of Ae. aegypti.
Keywords: Aedes aegypti; Kunitz-type inhibitor; digestive enzyme; insecticide; molecular docking; protease.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures




Similar articles
-
Fusarium begoniae metabolites: a promising larvicidal, pupicidal potential, histopathological alterations and detoxifications enzyme profiles of medically important mosquito vector Aedes aegypti, Culex quinquefasciatus and Anopheles stephensi.3 Biotech. 2024 Oct;14(10):226. doi: 10.1007/s13205-024-04061-z. Epub 2024 Sep 10. 3 Biotech. 2024. PMID: 39263325
-
Impact of physicochemical parameters of Aedes aegypti breeding habitats on mosquito productivity and the size of emerged adult mosquitoes in Ouagadougou City, Burkina Faso.Parasit Vectors. 2022 Dec 20;15(1):478. doi: 10.1186/s13071-022-05558-3. Parasit Vectors. 2022. PMID: 36539816 Free PMC article.
-
Natural vertical transmission of dengue virus in Aedes aegypti and Aedes albopictus: a systematic review.Parasit Vectors. 2018 Feb 1;11(1):77. doi: 10.1186/s13071-018-2643-9. Parasit Vectors. 2018. PMID: 29391071 Free PMC article.
-
Effects of desiccation stress on adult female longevity in Aedes aegypti and Ae. albopictus (Diptera: Culicidae): results of a systematic review and pooled survival analysis.Parasit Vectors. 2018 Apr 25;11(1):267. doi: 10.1186/s13071-018-2808-6. Parasit Vectors. 2018. PMID: 29695282 Free PMC article.
-
Larvicidal Activity of Essential Oil, Hydrolate, and Aqueous Extract from Leaves of Myrciaria floribunda Against Aedes Aegypti.Molecules. 2025 Jul 25;30(15):3116. doi: 10.3390/molecules30153116. Molecules. 2025. PMID: 40807291 Free PMC article.
References
-
- WHO – World Health Organization. Updated WHO Guidance for Controlling Vector-Borne Diseases Through Indoor Residual Spraying. 2024. [(accessed on 5 August 2024)]. Available online: https://www.who.int/news/item/15-02-2024-updated-who-guidance-for-contro....
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

