Twice as Effective? Pressurized Intra-Thoracic Aerosol Chemotherapy: New Frontiers in Pleural Mesothelioma
- PMID: 40559230
- PMCID: PMC12195480
- DOI: 10.3390/medsci13020072
Twice as Effective? Pressurized Intra-Thoracic Aerosol Chemotherapy: New Frontiers in Pleural Mesothelioma
Abstract
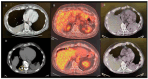
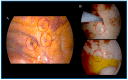
Pressurized intra-thoracic aerosol chemotherapy (PITAC) is a novel and promising strategy for the treatment of malignant pleural effusion (MPE). PITAC enables effective pleurodesis while potentially exerting an antineoplastic effect by delivering chemotherapeutic agents as a therapeutic aerosol into the thoracic cavity via a nebulizer. Our preliminary study involved nine patients with unresectable pleural mesothelioma (PM) treated with PITAC. Among them, one case was particularly emblematic for demonstrating notable oncological improvements in addition to well-known palliative benefits. This patient underwent two PITAC procedures, one year apart, without perioperative complications. Redo pleural biopsies from both previous and new sites revealed only fibrous tissue and inflammatory cells, with no evidence of malignancy. Beyond achieving pleurodesis, PITAC-by combining cytotoxic and sclerosing effects-may offer effective local antineoplastic control and represent a promising avenue for enhancing loco-regional therapy in PM.
Keywords: local antineoplastic control; malignant pleural effusion; mesothelioma; palliative care; pleural mesothelioma; pressurized intra-thoracic aerosol chemotherapy (PITAC).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Marinaccio A., Binazzi A., Marzio D.D., Scarselli A., Verardo M., Mirabelli D., Gennaro V., Mensi C., Riboldi L., Merler E., et al. Pleural malignant mesothelioma epidemic: Incidence, modalities of asbestos exposure and occupations involved from the Italian National Register. Int. J. Cancer. 2012;130:2146–2154. doi: 10.1002/ijc.26229. - DOI - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

