Evaluation of the Antileishmanial Activity of Some Benzimidazole Derivatives Using In Vitro and In Silico Techniques
- PMID: 40559787
- PMCID: PMC12197429
- DOI: 10.3390/vetsci12060550
Evaluation of the Antileishmanial Activity of Some Benzimidazole Derivatives Using In Vitro and In Silico Techniques
Abstract
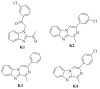
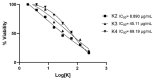
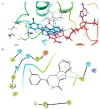
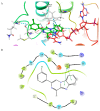
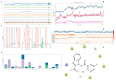
Benzimidazole derivatives are well known for their anthelmintic activity. Investigating the potential efficacy of new derivatives of this class against various parasites is essential to identify novel drug candidates. For this purpose, an in-house molecular database was screened, and four benzimidazole-based molecules were chosen to evaluate antiprotozoal activity. The compounds (K1-K4) had been previously synthesized through a four-step procedure. The potential in vitro cytotoxic properties of the compounds were assessed against the Leishmania (L.) major strain and L929 mouse fibroblast cells. The tests indicated that K1 (3-Cl phenyl) demonstrated an antileishmanial effect (IC50 = 0.6787 µg/mL) and cytotoxicity at elevated concentrations (CC50 = 250 µg/mL) in healthy cells. These findings were comparable to those of AmpB. The antileishmanial activity values were determined as follows: K2; 8.89 µg/mL, K3; 45.11 µg/mL, K4; and 69.19 µg/mL. The CC50 values were determined as follows: K2, 63 µg/mL; K3; 0.56 µg/mL; and K4, 292 µg/mL. Molecular docking and dynamic simulations were conducted to elucidate the potential mechanisms of action of the test substances. In silico investigations indicated interactions between the compounds and the active site of pteridine reductase 1 (PTR1), which is a biosynthetic enzyme essential for parasite proliferation. N-alkyl benzimidazole-based compounds exhibit potential inhibitory activity against L. (L.) major promastigotes. Therefore, these findings suggest that in vivo evaluation is warranted, and structural modifications may lead to the identification of more effective antileishmanial agents.
Keywords: Leishmania major; antileismanial activity; benzimidazole derivates; in silico; in vitro; pteridine reductase 1.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Prunus amygdalus var. amara seed extract enhances the antileishmanial activity of miltefosine.BMC Complement Med Ther. 2025 Jul 16;25(1):273. doi: 10.1186/s12906-025-04958-z. BMC Complement Med Ther. 2025. PMID: 40670989 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
In Vitro Anti-Leishmanial Activity and Molecular Docking of Eugenol as a Potential Agent Against Leishmania Major.Acta Parasitol. 2025 Jul 7;70(4):147. doi: 10.1007/s11686-025-01089-w. Acta Parasitol. 2025. PMID: 40622619
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
-
Inhaled mannitol for cystic fibrosis.Cochrane Database Syst Rev. 2018 Feb 9;2(2):CD008649. doi: 10.1002/14651858.CD008649.pub3. Cochrane Database Syst Rev. 2018. Update in: Cochrane Database Syst Rev. 2020 May 1;5:CD008649. doi: 10.1002/14651858.CD008649.pub4. PMID: 29424930 Free PMC article. Updated.
References
-
- World Health Organization (WHO) Leishmaniasis. [(accessed on 27 January 2025)]. Available online: https://www.who.int/news-room/fact-sheets/detail/leishmaniasis.
-
- World Health Organization (WHO) Fourteenth Meeting of the Strategic and Technical Advisory Group for Neglected Tropical Diseases, 22–24 June 2021. World Health Organization; Geneva, Switzerland: 2021.
LinkOut - more resources
Full Text Sources

