Functionalized Nitrobenzothiadiazoles as Embedded Fluorescent Probes
- PMID: 40560764
- PMCID: PMC12291599
- DOI: 10.1021/acs.joc.5c00700
Functionalized Nitrobenzothiadiazoles as Embedded Fluorescent Probes
Abstract
Described herein is the synthesis and photophysical characterization of novel dipeptidomimetic fluorogenic probes. The new dipeptidomimetic cassette is based on the environment-sensitive fluorophore nitrobenzothiadiazole (NBTD) and is designed to be embedded within a polypeptide backbone, not attached as part of a conformationally unrestrained amino acid side chain. Compounds 1-6 were prepared to investigate the effect of introducing a methyl group at positions 5 and 6 of the nitrobenzothiadiazole scaffold on their photophysical properties. Additionally, we investigated the effect of N-methylation of the 4-amino group on their chemical and photophysical properties. The nitrobenzothiadiazole scaffold was functionalized with amino- and carboxy-termini, resulting in a conformationally restricted dipeptidomimetic scaffold (NBTD-Gly) that can potentially be embedded within peptides or proteins of interest and used as a biophysical tool for studying protein structure and function. Thus, we investigated the synthesis and photophysical properties of functionalized nitrobenzothiadiazoles (1-11). The amino-terminus of the dipeptidomimetic scaffold (NBTD-Gly) was introduced at the benzylic position of the benzothiadiazole core using Gabriel synthesis, followed by nitration. The carboxy terminus was introduced via nucleophilic aromatic substitution as part of the glycine moiety. Subsequent steps involved the removal of a phthaloyl protecting group and a condensation reaction to form protected dipeptidomimetic analogues 7-11.
Figures
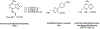
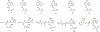
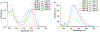


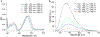
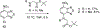
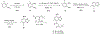

Similar articles
-
The effect of sample site and collection procedure on identification of SARS-CoV-2 infection.Cochrane Database Syst Rev. 2024 Dec 16;12(12):CD014780. doi: 10.1002/14651858.CD014780. Cochrane Database Syst Rev. 2024. PMID: 39679851 Free PMC article.
-
The Black Book of Psychotropic Dosing and Monitoring.Psychopharmacol Bull. 2024 Jul 8;54(3):8-59. Psychopharmacol Bull. 2024. PMID: 38993656 Free PMC article. Review.
-
Sexual Harassment and Prevention Training.2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. 2024 Mar 29. In: StatPearls [Internet]. Treasure Island (FL): StatPearls Publishing; 2025 Jan–. PMID: 36508513 Free Books & Documents.
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
Gonadotropin-releasing hormone (GnRH) analogues for premenstrual syndrome (PMS).Cochrane Database Syst Rev. 2025 Jun 10;6(6):CD011330. doi: 10.1002/14651858.CD011330.pub2. Cochrane Database Syst Rev. 2025. PMID: 40492482 Review.
References
-
- Budiarta M; Streit M; Beliu G. Site-Specific Protein Labeling Strategies for Super-Resolution Microscopy. Curr. Opin. Chem. Biol 2024, 80, 102445. - PubMed
-
- Choi JH; Kim S; Kang OY; Choi SY; Hyun JY; Lee HS; Shin I. Selective Fluorescent Labeling of Cellular Proteins and its Biological Applications. Chem. Soc. Rev 2024, 53, 9446–9489. - PubMed
-
- Fan Y; Zhang F; Hao Y; Chen L; Zhou Q; Zeng H; Song Y; Guo Z; Peng X; Ge G. Fluorogenic Probes for Functional Imaging of Endoplasmic Reticulum-Resident Proteins: From Molecular Engineering to Biomedical Applications. Adv. Funct. Mater 2024, 35, 2416057.
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

