The influence of hydrogel stiffness on axonal regeneration after spinal cord injury
- PMID: 40560940
- PMCID: PMC12192028
- DOI: 10.1371/journal.pone.0325798
The influence of hydrogel stiffness on axonal regeneration after spinal cord injury
Abstract
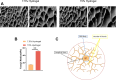
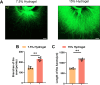
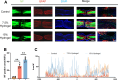
The core challenge in spinal cord injury(SCI) treatment is promoting axonal regeneration and forming new neural connections in damaged areas. However, mature CNS neurons have limited regenerative capacity, causing long-term dysfunction. Axonal regeneration involves elongating axons guided by growth cones, which sense and respond to external mechanical signals, integrating them into cytoskeleton reconstruction. After injury, growth cones experience altered mechanical forces due to changes in ECM stiffness. However, systematic studies on matrix stiffness's impact on axonal regeneration post-SCI remain insufficient. This study investigates the influence of hydrogel stiffness on axonal regeneration following SCI. Using gelatin methacryloyl (GelMA) hydrogels with varying stiffness levels, we cultured dorsal root ganglia (DRG) neurons in vitro and applied the hydrogels to a complete transection SCI mouse model. Results demonstrated that higher stiffness GelMA (15% w/v) significantly enhanced axonal extension and sensory functional recovery compared to lower stiffness (7.5% w/v). The study highlights the critical role of ECM stiffness in regulating axonal regeneration and suggests that optimizing hydrogel stiffness can promote neural regeneration and functional recovery after SCI. These findings provide valuable insights for developing therapeutic strategies in SCI treatment.
Copyright: © 2025 Deng et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures



Similar articles
-
PTEN Blocking Stimulates Corticospinal and Raphespinal Axonal Regeneration and Promotes Functional Recovery After Spinal Cord Injury.J Neuropathol Exp Neurol. 2021 Jan 20;80(2):169-181. doi: 10.1093/jnen/nlaa147. J Neuropathol Exp Neurol. 2021. PMID: 33367790
-
[Effect of removing microglia from spinal cord on nerve repair after spinal cord injury in mice].Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2025 Jun 15;39(6):754-761. doi: 10.7507/1002-1892.202503099. Zhongguo Xiu Fu Chong Jian Wai Ke Za Zhi. 2025. PMID: 40545466 Free PMC article. Chinese.
-
In vivo programming of adult pericytes aids axon regeneration by providing cellular bridges for SCI repair.Mol Ther. 2025 Aug 6;33(8):3968-3993. doi: 10.1016/j.ymthe.2025.04.020. Epub 2025 Apr 18. Mol Ther. 2025. PMID: 40253585
-
Efficacy of hydrogels for repair of traumatic spinal cord injuries: A systematic review and meta-analysis.J Biomed Mater Res B Appl Biomater. 2022 Jun;110(6):1460-1478. doi: 10.1002/jbm.b.34993. Epub 2021 Dec 13. J Biomed Mater Res B Appl Biomater. 2022. PMID: 34902215
-
Research progress on stiffness controllable scaffolds based on gelatin methacryloyl hydrogels for tissue repair and reconstruction.Int J Biol Macromol. 2025 Sep;321(Pt 3):146485. doi: 10.1016/j.ijbiomac.2025.146485. Epub 2025 Jul 31. Int J Biol Macromol. 2025. PMID: 40752703 Review.
References
-
- Choo AM, Liu J, Lam CK, Dvorak M, Tetzlaff W, Oxland TR. Contusion, dislocation, and distraction: primary hemorrhage and membrane permeability in distinct mechanisms of spinal cord injury. J Neurosurg Spine. 2007;6(3):255–66. - PubMed
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

