3D Printing in Nasal Reconstruction: Application-Based Evidence on What Works, When, and Why
- PMID: 40564153
- PMCID: PMC12190116
- DOI: 10.3390/biomedicines13061434
3D Printing in Nasal Reconstruction: Application-Based Evidence on What Works, When, and Why
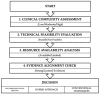
Abstract
Background: Nasal reconstruction requires a balance between aesthetic and functional restoration. Recent advances in three-dimensional (3D) printing have introduced new approaches to this field, enabling precise, patient-specific interventions. This review explores the applications, benefits, and challenges of integrating 3D printing in nasal reconstruction. Methods: A literature search was conducted using PubMed, Scopus, and Web of Science to identify studies on 3D printing in nasal reconstruction. Peer-reviewed articles and clinical trials were analyzed to assess the impact of 3D-printed models, implants, and bioengineered scaffolds. Results: 3D printing facilitates the creation of anatomical models, surgical guides, and implants, enhancing surgical precision and patient outcomes. Techniques such as stereolithography (SLA) and selective laser sintering (SLS) enable high-resolution, biocompatible constructs using materials like polylactic acid, titanium, and hydroxyapatite. Computational fluid dynamics (CFD) tools improve surgical planning by optimizing nasal airflow. Studies show that 3D-printed guides reduce operative time and improve symmetry. Emerging bioprinting techniques integrating autologous cells offer promise for tissue regeneration. Challenges and Future Directions: Challenges include high costs, imaging limitations, regulatory hurdles, and limited vascularization in bioprinted constructs. Future research should focus on integrating bioactive materials, artificial intelligence-assisted design, and regulatory standardization. Conclusions: 3D printing offers specific advantages in nasal reconstruction, improving precision and outcomes in selected cases. Addressing current limitations through technological and regulatory advancements will further its clinical integration, potentially enhancing reconstructive surgery techniques.
Keywords: 3D printing; bioprinting; computational modeling; nasal reconstruction; patient-specific implants; regenerative medicine.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Higgins J.P., Altman D.G., Gøtzsche P.C., Jüni P., Moher D., Oxman A.D., Savovic J., Schulz K.F., Weeks L., Sterne J.A. Cochrane Bias Methods Group; Cochrane Statistical Methods Group. The Cochrane Collaboration’s tool for assessing risk of bias in randomised trials. BMJ. 2011;343:d5928. doi: 10.1136/bmj.d5928. - DOI - PMC - PubMed
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

