Effects of Cholinergic and Opioid Antagonists on In Vitro Release of Met-Enkephalin, Somatostatin and Insulin-like Growth Factor-1 by and PENK Expression in Crop, Proventriculus and Duodenum of Newly Hatched Chickens
- PMID: 40564255
- PMCID: PMC12189355
- DOI: 10.3390/ani15121702
Effects of Cholinergic and Opioid Antagonists on In Vitro Release of Met-Enkephalin, Somatostatin and Insulin-like Growth Factor-1 by and PENK Expression in Crop, Proventriculus and Duodenum of Newly Hatched Chickens
Abstract
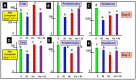
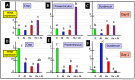
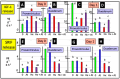
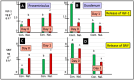
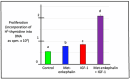
The gastrointestinal (GI) tract is under neural, endocrine and paracrine control. The release (basal and in the presence of either cholinergic and opioid antagonists) of Met-enkephalin, insulin-like growth factor 1 (IGF-1) and somatostatin (SRIF) was determined quantitatively in vitro using explants of the crop, proventriculus and duodenum from either day 0 or day 1 chicks. In addition, the effects of cholinergic and opioid antagonists on PENK gene expression were examined. Thus, the aim of this study was to determine the roles of cholinergic and opioid receptors in the GI tract in newly hatched chickens. Moreover, the effect of IGF-1 and Met-enkephalin on cell division in duodenal explants in vitro was determined. There was both the release of Met-enkephalin from, and PENK expression in, the explants of the crop, proventriculus and duodenum tissue. This is the first report of any neuropeptide(s) being synthesized in and/or released from the crop. In vitro release of Met-enkephalin, IGF-1 and SRIF from duodenal and proventriculus explants was influenced (p < 0.01) by either cholinergic or opioid antagonists; for instance, in the presence of atropine, decreases (p < 0.001) of 17.8% and 57.7% are seen, respectively, in Met-enkephalin release and PENK expression in crop explants from day 1 chicks. Moreover, in the presence of atropine, there were increases (p < 0.001) of 47.7% and 70.9% in IGF-1 release in proventriculus explants from, respectively, day 0 and day 1 chicks. Met-enkephalin and/or IGF-1 stimulated the cell division of duodenal explants in vitro. This is the first report of Met-enkephalin release and PENK expression in the avian crop and of the effects of cholinergic or opioid antagonists on these factors. It is also the first report of either cholinergic or opioid control of IGF-1 release in the periphery of any species. There were strong relationships (p < 0.05) between the release of Met-enkephalin and that of IGF-1 in the duodenum and between the release of SRIF and that of IGF-1 in the proventriculus. This is the first report of IGF-1 and Met-enkephalin stimulating (p < 0.001) the proliferation of duodenal cells and, together, exerting a synergist effect. It is concluded that the release of Met-enkephalin, IGF-1 and SRIF from foregut regions is under tonic cholinergic and opioid control.
Keywords: chicken; cholinergic; crop; duodenum; insulin-like growth factor-1; met-enkephalin; opioid; proventriculus; somatostatin (SRIF).
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Effects of cholinergic antagonists on ghrelin release and expression in the crop, proventriculus, and duodenum of newly hatched chickens.Front Physiol. 2025 May 2;16:1553474. doi: 10.3389/fphys.2025.1553474. eCollection 2025. Front Physiol. 2025. PMID: 40386621 Free PMC article.
-
Mu-opioid antagonists for opioid-induced bowel dysfunction in people with cancer and people receiving palliative care.Cochrane Database Syst Rev. 2018 Jun 5;6(6):CD006332. doi: 10.1002/14651858.CD006332.pub3. Cochrane Database Syst Rev. 2018. Update in: Cochrane Database Syst Rev. 2022 Sep 15;9:CD006332. doi: 10.1002/14651858.CD006332.pub4. PMID: 29869799 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2017 Dec 22;12(12):CD011535. doi: 10.1002/14651858.CD011535.pub2. Cochrane Database Syst Rev. 2017. Update in: Cochrane Database Syst Rev. 2020 Jan 9;1:CD011535. doi: 10.1002/14651858.CD011535.pub3. PMID: 29271481 Free PMC article. Updated.
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of topotecan for ovarian cancer.Health Technol Assess. 2001;5(28):1-110. doi: 10.3310/hta5280. Health Technol Assess. 2001. PMID: 11701100
References
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

