Alzheimer's Disease Prediction Using Fisher Mantis Optimization and Hybrid Deep Learning Models
- PMID: 40564769
- PMCID: PMC12191870
- DOI: 10.3390/diagnostics15121449
Alzheimer's Disease Prediction Using Fisher Mantis Optimization and Hybrid Deep Learning Models
Abstract
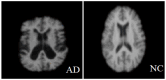
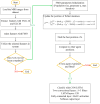
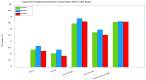
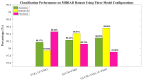
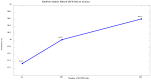
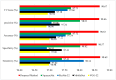
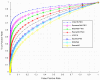
Background/Objectives: Alzheimer's disease (AD) is a progressive neurodegenerative disorder causing memory, cognitive, and behavioral decline. Early and accurate diagnosis is critical for timely treatment and management. This study proposes a novel hybrid deep learning framework, GLCM + VGG16 + FMO + CNN-LSTM, to improve AD diagnosis using MRI data. Methods: MRI images were preprocessed through normalization and noise reduction. Feature extraction combined texture features from the Gray-Level Co-occurrence Matrix (GLCM) and spatial features extracted from a pretrained VGG-16 network. Fisher Mantis Optimization (FMO) was employed for optimal feature selection. The selected features were classified using a CNN-LSTM model, capturing both spatial and temporal patterns. The MLP-LSTM model was included only for benchmarking purposes. The framework was evaluated on The ADNI and MIRIAD datasets. Results: The proposed method achieved 98.63% accuracy, 98.69% sensitivity, 98.66% precision, and 98.67% F1-score, outperforming CNN + SVM and 3D-CNN + BiLSTM by 2.4-3.5%. Comparative analysis confirmed FMO's superiority over other metaheuristics, such as PSO, ACO, GWO, and BFO. Sensitivity analysis demonstrated robustness to hyperparameter changes. Conclusions: The results confirm the efficacy and stability of the GLCM + VGG16 + FMO + CNN-LSTM model for accurate and early AD diagnosis, supporting its potential clinical application.
Keywords: Alzheimer’s disease diagnosis; CNN; Fisher Mantis Optimization algorithm; feature selection.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures

Similar articles
-
Predicting cognitive decline: Deep-learning reveals subtle brain changes in pre-MCI stage.J Prev Alzheimers Dis. 2025 May;12(5):100079. doi: 10.1016/j.tjpad.2025.100079. Epub 2025 Feb 6. J Prev Alzheimers Dis. 2025. PMID: 39920001 Free PMC article.
-
RESIGN: Alzheimer's Disease Detection Using Hybrid Deep Learning based Res-Inception Seg Network.Curr Alzheimer Res. 2025 Jun 18. doi: 10.2174/0115672050373821250612053943. Online ahead of print. Curr Alzheimer Res. 2025. PMID: 40539353
-
Biologically inspired hybrid model for Alzheimer's disease classification using structural MRI in the ADNI dataset.Front Artif Intell. 2025 Jun 19;8:1590599. doi: 10.3389/frai.2025.1590599. eCollection 2025. Front Artif Intell. 2025. PMID: 40612382 Free PMC article.
-
Selegiline for Alzheimer's disease.Cochrane Database Syst Rev. 2003;(1):CD000442. doi: 10.1002/14651858.CD000442. Cochrane Database Syst Rev. 2003. PMID: 12535396
-
Nimodipine for primary degenerative, mixed and vascular dementia.Cochrane Database Syst Rev. 2001;(1):CD000147. doi: 10.1002/14651858.CD000147. Cochrane Database Syst Rev. 2001. Update in: Cochrane Database Syst Rev. 2002;(3):CD000147. doi: 10.1002/14651858.CD000147. PMID: 11279679 Updated.
References
-
- Christina P. The State of the Art of Dementia Research: New Frontiers. Alzheimer’s Disease International; London, UK: 2018.
-
- Balagopalan A., Eyre B., Rudzicz F., Novikova J. To BERT or not to BERT: Comparing speech and language-based approaches for Alzheimer’s disease detection. arXiv. 20202008.01551
-
- Balagopalan A., Novikova J., Rudzicz F., Ghassemi M. The effect of heterogeneous data for Alzheimer’s disease detection from speech. arXiv. 20181811.12254
-
- Khosla A., Khandnor P., Chand T. A comparative analysis of signal processing and classification methods for different applications based on EEG signals. Biocybern. Biomed. Eng. 2020;40:649–690. doi: 10.1016/j.bbe.2020.02.002. - DOI
LinkOut - more resources
Full Text Sources

