Cold-Sensing TRP Channels and Temperature Preference Modulate Ovarian Development in the Model Organism Drosophila melanogaster
- PMID: 40565102
- PMCID: PMC12192652
- DOI: 10.3390/ijms26125638
Cold-Sensing TRP Channels and Temperature Preference Modulate Ovarian Development in the Model Organism Drosophila melanogaster
Abstract
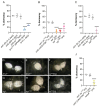
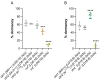
Temperature is perceived primarily via transient receptor potential (TRP) channels, which are integral to the molecular machinery sensing environmental and cellular signals. Functional evidence of TRP channels' involvement in regulating cold-induced developmental/reproductive responses remains scarce. Here, we show that mutations affecting cold-sensing TRP channels antagonize the reduction in ovarian development induced by low temperatures (reproductive dormancy) in Drosophila melanogaster. More specifically, mutants for brv1, trp, and trpl significantly lowered dormancy levels at 12 °C and exhibited well-developed oocytes characterized by advanced vitellogenesis. Similarly, functional knockouts for norpA, a gene encoding a phospholipase C acting downstream to Trp and Trpl, exhibited a reduced dormancy response, suggesting that Ca2+ signaling is key to relaying cold-sensing stimuli during dormancy induction and maintenance. Finally, mutants with an altered temperature preference (i.e., exhibiting impaired cold or warm avoidance) differentially responded to the cold, either lowering or increasing dormancy levels. In summary, our phenotypic analysis provides functional evidence of developmental/reproductive modulation by specific cold-sensing TRP channels in Drosophila melanogaster and indicates that temperature preference affects developmental processes.
Keywords: TRP channels; ovarian development; reproductive dormancy; temperature preference; temperature sensing.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Distinct TRP channels are required for warm and cool avoidance in Drosophila melanogaster.Proc Natl Acad Sci U S A. 2008 Sep 23;105(38):14668-73. doi: 10.1073/pnas.0805041105. Epub 2008 Sep 11. Proc Natl Acad Sci U S A. 2008. PMID: 18787131 Free PMC article.
-
Temperature sensitivity of spreading depolarization in the CNS of Drosophila melanogaster.Am J Physiol Regul Integr Comp Physiol. 2025 Jul 1;329(1):R134-R149. doi: 10.1152/ajpregu.00040.2025. Epub 2025 May 20. Am J Physiol Regul Integr Comp Physiol. 2025. PMID: 40392029
-
Exploiting TRP channel diversity in insects: a pathway to next-generation pest management.Arch Toxicol. 2025 Jun;99(6):2277-2297. doi: 10.1007/s00204-025-04012-4. Epub 2025 Mar 8. Arch Toxicol. 2025. PMID: 40056168 Review.
-
Impact of residual disease as a prognostic factor for survival in women with advanced epithelial ovarian cancer after primary surgery.Cochrane Database Syst Rev. 2022 Sep 26;9(9):CD015048. doi: 10.1002/14651858.CD015048.pub2. Cochrane Database Syst Rev. 2022. PMID: 36161421 Free PMC article.
-
Aromatase inhibitors (letrozole) for subfertile women with polycystic ovary syndrome.Cochrane Database Syst Rev. 2018 May 24;5(5):CD010287. doi: 10.1002/14651858.CD010287.pub3. Cochrane Database Syst Rev. 2018. Update in: Cochrane Database Syst Rev. 2022 Sep 27;9:CD010287. doi: 10.1002/14651858.CD010287.pub4. PMID: 29797697 Free PMC article. Updated.
References
MeSH terms
Substances
Grants and funding
- NA/CaRiPaRo
- Junior Research Fellowship/Department of Biology at the University of Padua
- Postdoctoral fellowship (Project N° 101109842)/Marie Sklodowska-Curie Actions
- Comparative Insect Chronobiology (CINCHRON), Marie Sklodowska-Curie Initial Training Network/EU Horizon 2020
- project 101169474 - INCITE. Horizon-MSCA-2023-DN-01/EU Horizon 2020
LinkOut - more resources
Full Text Sources
Miscellaneous

