Evaluating the Antitumor Potential of Cannabichromene, Cannabigerol, and Related Compounds from Cannabis sativa and Piper nigrum Against Malignant Glioma: An In Silico to In Vitro Approach
- PMID: 40565152
- PMCID: PMC12192948
- DOI: 10.3390/ijms26125688
Evaluating the Antitumor Potential of Cannabichromene, Cannabigerol, and Related Compounds from Cannabis sativa and Piper nigrum Against Malignant Glioma: An In Silico to In Vitro Approach
Abstract
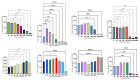
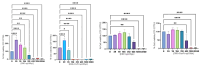
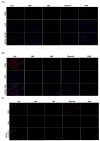
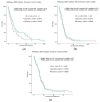
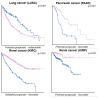
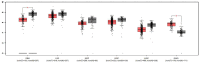
Malignant gliomas, including glioblastoma multiforme (GBM), are highly aggressive brain tumors with a poor prognosis and limited treatment options. This study investigates the antitumor potential of bioactive compounds derived from Cannabis sativa and Piper nigrum using molecular docking, cell viability assays, and transcriptomic and expression analyses from public databases in humans and cell lines. Cannabichromene (CBC), cannabigerol (CBG), cannabidiol (CBD), and Piper nigrum derivates exhibited strong binding affinities relative to glioblastoma-associated targets GPR55 and PINK1. In vitro analyses demonstrated their cytotoxic effects on glioblastoma cell lines (U87MG, T98G, and CCF-STTG1), as well as on neuroblastoma (SH-SY5Y) and oligodendroglial (MO3.13) cell lines, revealing interactions among these compounds. The differential expression of GPR55 and PINK1 in tumor versus normal tissues further supports their potential as biomarkers and therapeutic targets. These findings provide a basis for the development of novel therapies and suggest unexplored molecular pathways for the treatment of malignant glioma.
Keywords: CCF-STTG1; Cannabis sativa; GPR55; PINK1; Piper nigrum; T98G; U87MG; cannabichromene; cannabidiol; cannabidiolic acid (CBDA); cannabigerol; glioblastoma; piperine.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures



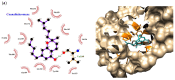
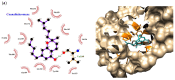
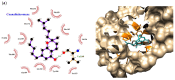
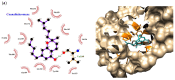




References
-
- Kurdi M., Alkhotani A., Alsinani T., Alkhayyat S., Katib Y., Jastaniah Z., Sabbagh A.J., Butt N.S., Toonsi F.A., Alharbi M., et al. Effects of Radiotherapy Alone Versus Concomitant Radiotherapy with Temozolomide Chemotherapy on the Outcome of IDH-Wildtype Glioblastoma Patients. Clin. Oncol. 2025;38:103741. doi: 10.1016/j.clon.2024.103741. - DOI - PubMed
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical
Research Materials
Miscellaneous

