Retinal Autophagy for Sustaining Retinal Integrity as a Proof of Concept for Age-Related Macular Degeneration
- PMID: 40565235
- PMCID: PMC12193083
- DOI: 10.3390/ijms26125773
Retinal Autophagy for Sustaining Retinal Integrity as a Proof of Concept for Age-Related Macular Degeneration
Abstract
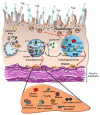
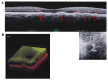
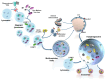
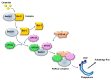
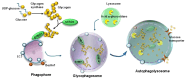
Current evidence indicates that most types of autophagy represent a pivot in promoting retinal integrity. In healthy conditions, autophagy acts on multiple pathways, which are fundamental for the biochemistry and the fine structure of the retina. Autophagy is essential in granting visual processes. On the other hand, autophagy dysfunction characterizes several retinal disorders. This is mostly evident in age-related macular degeneration (AMD), which represents the most common degenerative disease leading to blindness. The involvement of autophagy in AMD is documented in vitro and in vivo experiments, and it is strongly suggested by clinical findings in humans. The present manuscript provides an overview of the specific types of autophagy, which prevail in the retina and their alterations in retinal degeneration with an emphasis on AMD. The dysfunction of specific autophagy steps was analyzed in relation to hallmarks of AMD pathology and symptoms. An extended session of the manuscript analyzes the connection between altered autophagy and cell pathology within retinal pigment epithelium, as well as the site and structure of extracellular aggregates named drusen. The significance of the drusen in relation to visual function is discussed in the light of the role of autophagy in regulating key steps of phototransduction.
Keywords: autophagoproteasome; drusen; lipophagy; lysosome; mitophagy; proteasome; pseudodrusen; retinal degeneration; retinal neurovascular unit; retinal pigment epithelium.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures







Similar articles
-
The role of secretory autophagy and exosomes in the accumulation of drusen during the development of age-related macular degeneration (AMD).Ageing Res Rev. 2025 Aug;110:102796. doi: 10.1016/j.arr.2025.102796. Epub 2025 Jun 3. Ageing Res Rev. 2025. PMID: 40473035 Review.
-
Surgery for cataracts in people with age-related macular degeneration.Cochrane Database Syst Rev. 2017 Feb 16;2(2):CD006757. doi: 10.1002/14651858.CD006757.pub4. Cochrane Database Syst Rev. 2017. PMID: 28206671 Free PMC article.
-
Recycling the recyclers: lysophagy emerges as a new pharmacological target for retinal degeneration.Autophagy. 2024 Nov;20(11):2589-2590. doi: 10.1080/15548627.2024.2391726. Epub 2024 Aug 17. Autophagy. 2024. PMID: 39133039 Free PMC article.
-
Age-Associated Decline in Autophagy Pathways in the Retinal Pigment Epithelium and Protective Effects of Topical Trehalose in Light-Induced Outer Retinal Degeneration in Mice.Aging Cell. 2025 Jul;24(7):e70081. doi: 10.1111/acel.70081. Epub 2025 Apr 28. Aging Cell. 2025. PMID: 40293407 Free PMC article.
-
A longitudinal study of the 5xFAD mouse retina delineates Amyloid beta (Aβ)-mediated retinal pathology from age-related changes.Alzheimers Res Ther. 2025 Jun 19;17(1):136. doi: 10.1186/s13195-025-01784-w. Alzheimers Res Ther. 2025. PMID: 40537856 Free PMC article.
References
Publication types
MeSH terms
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

