Discovery of Novel Phenolic Compounds from Eutypa lata Through OSMAC Approach: Structural Elucidation and Antibiotic Potential
- PMID: 40565239
- PMCID: PMC12193298
- DOI: 10.3390/ijms26125774
Discovery of Novel Phenolic Compounds from Eutypa lata Through OSMAC Approach: Structural Elucidation and Antibiotic Potential
Abstract
Among grapevine trunk diseases, Eutypa dieback, caused by the fungus Eutypa lata, is one of the most critical ones, due to its widespread infection in vineyards and the lack of effective treatments. This fungus is a vascular pathogen that enters grapevines through pruning wounds. The infection process is associated with phytotoxic metabolites produced by the fungus, and as such, the identification of new metabolites from different culture conditions and broths could provide valuable insights into the fungus's enzymatic system and help its control. For the purposes of this study, the OSMAC (one strain, many compounds) approach was applied to investigate the secondary metabolism of E. lata strain 311 isolated from Vitis vinifera plants in Spain. A total of twenty metabolites were isolated, including five reported for the first time from E. lata and four that are newly identified compounds in the literature: eulatagalactoside A, (R)-2-(4'-hydroxy-3'-methylbut-1'-yn-1'-yl)-4-(hydroxymethyl)phenol, (S)-7-hydroxymethyl-3-methyl-2,3-dihydro-1-benzoxepin-3-ol, and (3aR,4S,5R,7aS)-4,5-dihydroxy-6-((R)-3'-methylbuta-1',3'-dien-1'-ylidene)hexahydrobenzo[d][1,3]dioxol-2-one. These compounds were extracted from fermentation broths using silica gel column chromatography and high-performance liquid chromatography (HPLC). Their structures were elucidated through extensive 1D and 2D NMR spectroscopy, along with high-resolution electrospray ionization mass spectrometry (HRESIMS). Compounds were evaluated for phytotoxicity against Phaseolus vulgaris, with only eulatagalactoside A producing white spots after 48 h. Additionally, the antibacterial activity against Escherichia coli, Staphylococcus aureus, and Klebsiella pneumoniae of selected compounds was tested. The compounds (R)-2-(4'-hydroxy-3'-methylbut-1'-yn-1'-yl)-4-(hydroxymethyl)phenol and (S)-7-(hydroxymethyl)-3-methyl-2,3-dihydrobenzo[b]oxepin-3-ol showed the most significant antimicrobial activity against Gram-positive bacteria, inhibiting S. aureus by over 75%, with IC50 values of 511.4 µg/mL and 617.9 µg/mL, respectively.
Keywords: Eutypa; GTDs; phytopathogen; secondary metabolites; toxins.
Conflict of interest statement
Author Manuel Delgado is employed by the company González Byass, S.A. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures
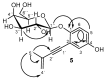
 HMBC spectra correlations for 5.
HMBC spectra correlations for 5.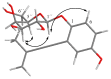
 NOESY spectra correlations for 5.
NOESY spectra correlations for 5.
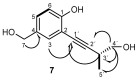
 HMBC and
HMBC and  COSY spectra correlations for 7.
COSY spectra correlations for 7.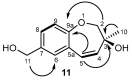
 HMBC and
HMBC and  COSY spectra correlations for 11.
COSY spectra correlations for 11.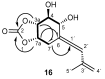
 HMBC and
HMBC and  COSY spectra correlations for 16.
COSY spectra correlations for 16.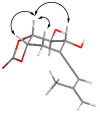
 Selected NOE interactions and correlations exhibited by 16.
Selected NOE interactions and correlations exhibited by 16.
References
-
- Fontaine F., Gramaje D., Armengol J., Smart R., Nagy Z.A., Borgo M., Rego C., Corio-Costet M.-F. Grapevine Trunk Diseases. A Review. International Organisation of Vine and Wine (OIV); Paris, France: 2016. p. 25.
-
- Kenfaoui J., Radouane N., Mennani M., Tahiri A., El Ghadraoui L., Belabess Z., Fontaine F., El Hamss H., Amiri S., Lahlali R., et al. A Panoramic View on Grapevine Trunk Diseases Threats: Case of Eutypa Dieback, Botryosphaeria Dieback, and Esca Disease. J. Fungi. 2022;8:595. doi: 10.3390/jof8060595. - DOI - PMC - PubMed
-
- Lorrain B., Ky I., Pasquier G., Jourdes M., Dubrana L.G., Gény L., Rey P., Donèche B., Teissedre P.L. Effect of Esca Disease on the Phenolic and Sensory Attributes of Cabernet Sauvignon Grapes, Musts and Wines. Aust. J. Grape Wine Res. 2012;18:64–72. doi: 10.1111/j.1755-0238.2011.00172.x. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

