Differences in Exercise Performance in Fontan Patients with Extracardiac Conduit and Lateral Tunnel: A FORCE Fontan Registry Study
- PMID: 40565813
- PMCID: PMC12193827
- DOI: 10.3390/jcm14124067
Differences in Exercise Performance in Fontan Patients with Extracardiac Conduit and Lateral Tunnel: A FORCE Fontan Registry Study
Abstract
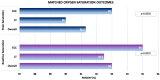
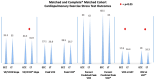
Background: To explore the differences in exercise capacity between the extracardiac conduit (ECC) and lateral tunnel (LT) Fontan. Methods: 2169 patients (36% LT (n = 774); 64% ECC (n = 1395)) underwent a Fontan operation between 2000 to 2023 in a multi-institutional Fontan registry. LT patients were age-matched to ECC patients, and cardiopulmonary exercise test (CPET) results were compared. Following age-matching and exclusion of those without CPET data, 470 patients emerged with 235 LT and 235 ECC patients. Results: ECC achieved higher peak heart rates (174 vs. 169 bpm, p = 0.0008) and heart rates at ventilatory anaerobic threshold (VAT) (130 vs. 119 bpm p = 0.0005). Oxygen saturations at peak (93.0 vs. 90.0%, p = 0.0003) and baseline (95 vs. 92.5%, p < 0.0001) were higher in the ECC group. The VO2 at VAT was higher in the ECC (17.8 vs. 16.4 mL/kg/min p = 0.0123). Baseline pre-exercise heart rate, peak oxygen pulse, VE/VCO2 slope, peak VO2, peak % of predicted VO2, peak work rate, and peak % of predicted work rate were similar (all, p > 0.05). Notably, less than 35% of the cohort had a documented CPET. Conclusions: We found that the ECC performed statistically better on many parameters of exercise capacity, including the ability to increase heart rate, have higher peak and baseline saturations, and to achieve superior VO2 at VAT. However, the magnitude of difference was small, suggesting that the translational value into the clinical realm may be limited. With a minority of the registry patients having CPET completed, this illuminates the need for the implementation of CPET surveillance for Fontan patients.
Keywords: Fontan; exercise testing.
Conflict of interest statement
Drs. Alsaied, Files, and Rathod have research grant support from Mezzion Pharmaceuticals as PIs of the Fontan Udenafil Exercise Longitudinal Assessment-2 (FUEL-2) Trial (NCT # NCT05918211). No other relevant conflicts.
Figures

References
-
- Downing T.E., Allen K.Y., Glatz A.C., Rogers L.S., Ravishankar C., Rychik J., Faerber J.A., Fuller S., Montenegro L.M., Steven J.M., et al. Long-term survival after the Fontan operation: Twenty years of experience at a single center. J. Thorac. Cardiovasc. Surg. 2017;154:243–253.e2. doi: 10.1016/j.jtcvs.2017.01.056. - DOI - PubMed
-
- Iyengar A.J., Winlaw D.S., Grigg L.E., Celermajer D.S., D’udekem Y. Redefining expectations of long-term survival after the Fontan procedure: Twenty-five years of follow-up from the entire population of Australia and New Zealand. Circulation. 2014;130((Suppl. 1)):S32–S38. doi: 10.1016/j.hlc.2014.12.030. - DOI - PubMed
-
- Mery C.M., De León L.E., Trujillo-Diaz D., Ocampo E.C., Dickerson H.A., Zhu H., Adachi I., Heinle J.S., Fraser C.D., Ermis P.R. Contemporary Outcomes of the Fontan Operation: A Large Single-Institution Cohort. Ann. Thorac. Surg. 2019;108:1439–1446. doi: 10.1016/j.athoracsur.2019.05.039. - DOI - PubMed
LinkOut - more resources
Full Text Sources

