Increased sensitivity of primary aniridia limbal stromal cells to travoprost, leading to elevated migration and MMP-9 protein levels, in vitro
- PMID: 40570009
- PMCID: PMC12200743
- DOI: 10.1371/journal.pone.0326967
Increased sensitivity of primary aniridia limbal stromal cells to travoprost, leading to elevated migration and MMP-9 protein levels, in vitro
Abstract
Purpose: In congenital aniridia, not only limbal epithelial cells but also limbal stromal cells may contribute to the development of aniridia associated keratopathy (AAK). Secondary glaucoma affects 50-75% of patients with congenital aniridia, and prostaglandin analogs are commonly used for conservative treatment. This study aimed to explore the effect of travoprost on corneal limbal stromal cells from healthy (LSCs) and congenital aniridia subjects (AN-LSCs), in vitro.
Materials and methods: Cells were extracted from aniridia (AN-LSCs) (n=7) and healthy donors (LSCs) (n=7). In culture, the cells were treated with travoprost at concentrations ranging from 0.039-40 μg/mL for 20 minutes. Cell viability, proliferation and migration were determined to assess the effect of travoprost on AN-LSCs and LSCs. Analysis of inflammation-, retinoic acid signaling-, and apoptosis-related genes and proteins was performed using qPCR, Western blot, and ELISA. One-way ANOVA was used to analyze cell viability and proliferation. The Mann-Whitney test was applied to compare between-group differences, while the Friedman test was used to assess within-group differences.
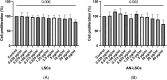
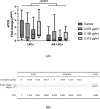
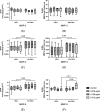
Results: Both in LSCs and AN-LSCs, travoprost treatment at 0.078 μg/mL and higher concentrations significantly reduced cell viability (p≤0.033; p<0.001) and proliferation decreased both in LSCs and AN-LSCs at 40 μg/mL travoprost concentration (p=0.006; p=0.002). At 6 and 12 hours, 0.313 μg/mL travoprost significantly increased the migration rate of AN-LSCs (p=0.021; p=0.021). AN-LSCs displayed lower PAX6 and JNK (MAPK8) mRNA (p<0.001) but higher MMP-3, MMP-9, ADH7, FABP5 and VEGFA mRNA levels (p≤0.037) than LSCs. PTGFR and JNK mRNA levels, MMP9 and ADH7 protein levels increased significantly in AN-LSCs after 0.313 μg/mL travoprost treatment (p≤0.039), while NF-κB and ADH7 protein levels decreased significantly in LSCs using 0.313 μg/mL travoprost (p=0.039; p<0.001).
Conclusions: Travoprost may affect viability, proliferation, and migration of both LSCs and AN-LSCs, with AN-LSCs exhibiting greater sensitivity than LSCs. Additionally, travoprost may regulate MMP-9 expression in AN-LSCs via the JNK signaling pathway. Furthermore, in AN-LSCs, travoprost treatment does not lead to a decrease in NF-κB and ADH7 protein levels.
Copyright: © 2025 Li et al. This is an open access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original author and source are credited.
Conflict of interest statement
The authors have declared that no competing interests exist.
Figures
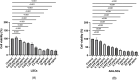





Similar articles
-
The effect of travoprost on primary human limbal epithelial cells and the siRNA-based aniridia limbal epithelial cell model, in vitro.PLoS One. 2025 Aug 19;20(8):e0330492. doi: 10.1371/journal.pone.0330492. eCollection 2025. PLoS One. 2025. PMID: 40828815 Free PMC article.
-
Altered Bcl-2/Caspase signaling and hypoxia-induced apoptosis in primary human aniridia limbal stromal cells, in CoCl2 mediated hypoxic stress, in vitro.PLoS One. 2025 Jul 10;20(7):e0328157. doi: 10.1371/journal.pone.0328157. eCollection 2025. PLoS One. 2025. PMID: 40638678 Free PMC article.
-
Effect of retinoic acid treatment on the retinoic acid signaling pathway in a human siRNA-based aniridia limbal epithelial cell model, in vitro.PLoS One. 2025 Jun 18;20(6):e0324946. doi: 10.1371/journal.pone.0324946. eCollection 2025. PLoS One. 2025. PMID: 40531840 Free PMC article.
-
Intravenous magnesium sulphate and sotalol for prevention of atrial fibrillation after coronary artery bypass surgery: a systematic review and economic evaluation.Health Technol Assess. 2008 Jun;12(28):iii-iv, ix-95. doi: 10.3310/hta12280. Health Technol Assess. 2008. PMID: 18547499
-
Levetiracetam add-on for drug-resistant focal epilepsy: an updated Cochrane Review.Cochrane Database Syst Rev. 2012 Sep 12;2012(9):CD001901. doi: 10.1002/14651858.CD001901.pub2. Cochrane Database Syst Rev. 2012. PMID: 22972056 Free PMC article.
Cited by
-
The effect of travoprost on primary human limbal epithelial cells and the siRNA-based aniridia limbal epithelial cell model, in vitro.PLoS One. 2025 Aug 19;20(8):e0330492. doi: 10.1371/journal.pone.0330492. eCollection 2025. PLoS One. 2025. PMID: 40828815 Free PMC article.
-
Altered Bcl-2/Caspase signaling and hypoxia-induced apoptosis in primary human aniridia limbal stromal cells, in CoCl2 mediated hypoxic stress, in vitro.PLoS One. 2025 Jul 10;20(7):e0328157. doi: 10.1371/journal.pone.0328157. eCollection 2025. PLoS One. 2025. PMID: 40638678 Free PMC article.
References
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Research Materials
Miscellaneous

