Biodegradable Contact Lenses for Targeted Ocular Drug Delivery: Recent Advances, Clinical Applications, and Translational Perspectives
- PMID: 40572507
- PMCID: PMC12195844
- DOI: 10.3390/molecules30122542
Biodegradable Contact Lenses for Targeted Ocular Drug Delivery: Recent Advances, Clinical Applications, and Translational Perspectives
Abstract
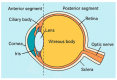
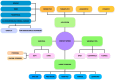
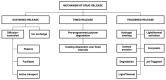
Ocular drug delivery presents a persistent clinical challenge due to the protective anatomical structure of the eye, physiological barriers such as reflex blinking, and continuous tear fluid turnover. These factors significantly limit the bioavailability of topically applied medications, reducing the therapeutic effectiveness of conventional formulations, such as eye drops, ointments, and suspensions, particularly in the management of chronic ocular disorders, including dry eye syndrome, diabetic retinopathy, and age-related macular degeneration. Drug-eluting contact lenses (DECLs) offer a promising alternative, enabling sustained, localized, and controlled drug release directly at the ocular surface. While several reviews have addressed contact lenses as drug delivery platforms, this work provides a distinct perspective by focusing specifically on biodegradable polymer-based systems. Emphasis is placed on recent advances in the design and fabrication of DECLs using natural and synthetic biodegradable polymers, which offer superior biocompatibility, customizable degradation kinetics, and the capacity for programmable drug release. This review discusses the selection criteria for polymer matrices, strategies for drug incorporation, and key factors influencing release profiles. Moreover, this study highlights innovative methodologies and therapeutic approaches that differentiate it from the existing literature, providing a timely and comprehensive resource for researchers developing next-generation polymeric ocular drug delivery systems.
Keywords: biodegradable polymers; drug-eluting contact lenses (DECLs); ophthalmic biomaterials; sustained drug release.
Conflict of interest statement
The author declares no conflicts of interest.
Figures

Similar articles
-
Innovative solid lipid nanoparticle formulations for ocular therapeutics and related patents: A systematic review.J Control Release. 2025 Sep 10;385:113992. doi: 10.1016/j.jconrel.2025.113992. Epub 2025 Jun 27. J Control Release. 2025. PMID: 40582641 Review.
-
Interventions to slow progression of myopia in children.Cochrane Database Syst Rev. 2011 Dec 7;(12):CD004916. doi: 10.1002/14651858.CD004916.pub3. Cochrane Database Syst Rev. 2011. Update in: Cochrane Database Syst Rev. 2020 Jan 13;1:CD004916. doi: 10.1002/14651858.CD004916.pub4. PMID: 22161388 Free PMC article. Updated.
-
Drug-eluting contact lenses: Progress, challenges, and prospects.Biointerphases. 2024 Jul 1;19(4):040801. doi: 10.1116/6.0003612. Biointerphases. 2024. PMID: 38984804 Review.
-
Contact Lenses in Therapeutic Care: A Comprehensive Review of Past Innovations, Present Applications, and Future Directions.Adv Exp Med Biol. 2025;1483:171-186. doi: 10.1007/5584_2024_842. Adv Exp Med Biol. 2025. PMID: 39841382 Review.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
References
-
- Efron N. Contact Lens Practice. Elsevier; Amsterdam, The Netherlands: 2024. Rigid Lens Materials; pp. 122–130.e1. - DOI
Publication types
MeSH terms
Substances
LinkOut - more resources
Full Text Sources
Medical

