Understanding the Light-Driven Enhancement of CO2 Hydrogenation over Ru/TiO2 Catalysts
- PMID: 40572541
- PMCID: PMC12196449
- DOI: 10.3390/molecules30122577
Understanding the Light-Driven Enhancement of CO2 Hydrogenation over Ru/TiO2 Catalysts
Abstract
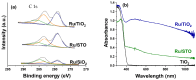
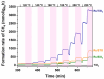
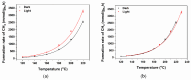
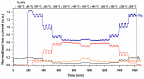
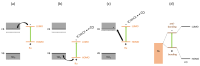
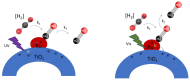
Ru/TiO2 catalysts are well known for their high activity in the hydrogenation of CO2 to CH4 (the Sabatier reaction). This activity is commonly attributed to strong metal-support interactions (SMSIs), associated with reducible oxide layers partly covering the Ru-metal particles. Moreover, isothermal rates of formation of CH4 can be significantly enhanced by the exposure of Ru/TiO2 to light of UV/visible wavelengths, even at relatively low intensities. In this study, we confirm the significant enhancement in the rate of formation of methane in the conversion of CO2, e.g., at 200 °C from ~1.2 mol gRu-1·h-1 to ~1.8 mol gRu-1·h-1 by UV/Vis illumination of a hydrogen-treated Ru/TiOx catalyst. The activation energy does not change upon illumination-the rate enhancement coincides with a temperature increase of approximately 10 °C in steady state (flow) conditions. In-situ DRIFT experiments, performed in batch mode, demonstrate that the Ru-CO absorption frequency is shifted and the intensity reduced by combined UV/Vis illumination in the temperature range of 200-350 °C, which is more significant than can be explained by temperature enhancement alone. Moreover, exposing the catalyst to either UV (predominantly exciting TiO2) or visible illumination (exclusively exciting Ru) at small intensities leads to very similar effects on Ru-CO IR intensities, formed in situ by exposure to CO2. This further confirms that the temperature increase is likely not the only explanation for the enhancement in the reaction rates. Rather, as corroborated by photophysical studies reported in the literature, we propose that illumination induces changes in the electron density of Ru partly covered by a thin layer of TiOx, lowering the CO coverage, and thus enhancing the methane formation rate upon illumination.
Keywords: CO coverage; CO2 hydrogenation; DRIFT spectroscopy; Ru/TiO2; charge transfer processes; heat; photothermal catalysis.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


References
-
- Zhang F., Li Y.-H., Qi M.-Y., Yamada Y.M.A., Anpo M., Tang Z.-R., Xu Y.-J. Photothermal catalytic CO2 reduction over nanomaterials. Chem Catal. 2021;1:272–297. doi: 10.1016/j.checat.2021.01.003. - DOI
-
- Yang X., Tan F., Wang D., Feng Q., Qiu D., Dang D., Wang X. Entrapping Ru nanoparticles into TiO2 nanotube: Insight into the confinement synergy on boosting pho-thermal CO2 methanation activity. Ceram. Int. 2021;47:27316–27323. doi: 10.1016/j.ceramint.2021.06.153. - DOI
-
- Kondratenko E.V., Mul G., Baltrusaitis J., Larrazábal G.O., Pérez-Ramírez J. Status and perspectives of CO2 conversion into fuels and chemicals by catalytic, photocatalytic and electrocatalytic processes. Energy Environ. Sci. 2013;6:3112–3135. doi: 10.1039/c3ee41272e. - DOI
-
- Wang C., Fang S., Xie S., Zheng Y., Hu Y.H. Thermo-photo catalytic CO2 hydrogenation over Ru/TiO2. J. Mater. Chem. A. 2020;8:7390–7394. doi: 10.1039/C9TA13275A. - DOI
Grants and funding
LinkOut - more resources
Full Text Sources
Medical

