Preparation of Biochars from Different Sources and Study on Their Phosphorus Adsorption Properties
- PMID: 40572596
- PMCID: PMC12195846
- DOI: 10.3390/molecules30122633
Preparation of Biochars from Different Sources and Study on Their Phosphorus Adsorption Properties
Abstract
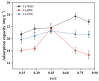
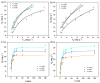
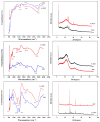
Biochar is a solid product generated through the pyrolysis of biomass materials under anaerobic or hypoxic conditions, and it is characterized by its strong adsorption capacity. To investigate the phosphorus adsorption performance of biochar derived from wheat straw, bamboo, and water hyacinth in wastewater, iron modification treatments were applied to these biochars, and the most effective modified biochar was identified. The physicochemical properties of the modified biochars were characterized using Fourier Transform Infrared Spectroscopy (FTIR), X-ray Diffraction (XRD), and scanning electron microscopy (SEM). The results showed that optimal modification was achieved with an iron-carbon mass ratio of 0.70 for wheat straw biochar (Fe-WBC) and 0.45 for both bamboo biochar (Fe-BBC) and water hyacinth biochar (Fe-HBC). The maximum phosphorus adsorption capacities of the three modified biochars were as follows: 31.76 mg g-1 (Fe-WBC) > 27.14 mg g-1 (Fe-HBC) > 25.31 mg g-1 (Fe-BBC). It was demonstrated that the adsorption behavior of Fe-BBC was predominantly multi-molecular layer adsorption, whereas the adsorption behavior of Fe-WBC and Fe-HBC was primarily monolayer adsorption. All three types of modified biochars reached adsorption equilibrium within 30 min, with Fe-WBC exhibiting the best adsorption performance. Analysis revealed that the modified biochars contained a large number of unsaturated C bonds and aromatic rings, indicating relatively stable structures. The surfaces of the modified biochars were rich in hydroxyl and carbonyl groups, which contributed to their strong adsorption properties. Post-modification analysis indicated that iron in the biochars predominantly existed in forms such as goethite (FeOOH) and hematite (Fe2O3). The iron content in each type of modified biochar constituted approximately 3.08% for Fe-WBC, 5.94% for Fe-BBC, and 5.68% for Fe-HBC relative to their total elemental composition. Overall, the iron-modified biochars employed in this study significantly enhanced the adsorption capacity and efficiency for phosphorus removal in wastewater.
Keywords: adsorption; biochar; iron oxide; modification; phosphorus.
Conflict of interest statement
Authors Yinlong Shao, Yongcan Jiang and Xianbiao Wang were employed by the company PowerChina Huadong Engineering Corporation Ltd. The remaining authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures

Similar articles
-
Adsorption Performance of Zn(II)-Based Coordination Polymer (ZnMOF) Reinforced Magnetic Activated Biochar (CmBC-Fe3O4@ZnMOF) Hybrid Composites.Water Environ Res. 2025 Jun;97(6):e70113. doi: 10.1002/wer.70113. Water Environ Res. 2025. PMID: 40542559 Free PMC article.
-
Tuning Electronic and Pore Structures of Biochar via Nitrogen and Magnesium Doping for Superior Methylene Blue Adsorption: Synergistic Mechanisms and Kinetic Analysis.ACS Omega. 2025 Jul 21;10(29):31679-31692. doi: 10.1021/acsomega.5c02636. eCollection 2025 Jul 29. ACS Omega. 2025. PMID: 40757343 Free PMC article.
-
Production and characterization of magnetic Biochar derived from pyrolysis of waste areca nut husk for removal of methylene blue dye from wastewater.Sci Rep. 2025 Jul 2;15(1):23209. doi: 10.1038/s41598-025-03359-z. Sci Rep. 2025. PMID: 40603323 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
Interventions for paracetamol (acetaminophen) overdose.Cochrane Database Syst Rev. 2018 Feb 23;2(2):CD003328. doi: 10.1002/14651858.CD003328.pub3. Cochrane Database Syst Rev. 2018. PMID: 29473717 Free PMC article.
References
-
- Qu J., Akindolie M.S., Feng Y., Jiang Z., Zhang G., Jiang Q., Deng F., Cao B., Zhang Y. One-pot hydrothermal synthesis of NaLa(CO3)2 decorated magnetic biochar for efficient phosphate removal from water: Kinetics, isotherms, thermodynamics, mechanisms and reusability exploration. Chem. Eng. J. 2020;394:124915. doi: 10.1016/j.cej.2020.124915. - DOI
-
- Cao J.S., Zhao W.Y., Wang S.N., Xu R.Z., Hao L.S., Sun W. Effects of Calcium on Phosphorus Recovery from Wastewater by Vivianite Crystallization: Interaction and Mechanism Analysis. J. Environ. Chem. Eng. 2023;11:9. doi: 10.1016/j.jece.2023.110506. - DOI
MeSH terms
Substances
Grants and funding
LinkOut - more resources
Full Text Sources

