Withaferin A Rescues Brain Network Dysfunction and Cognitive Deficits in a Mouse Model of Alzheimer's Disease
- PMID: 40573212
- PMCID: PMC12196360
- DOI: 10.3390/ph18060816
Withaferin A Rescues Brain Network Dysfunction and Cognitive Deficits in a Mouse Model of Alzheimer's Disease
Abstract
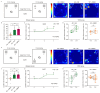
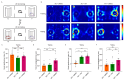
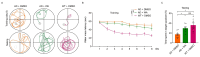
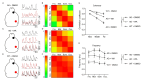
Background: Alzheimer's disease (AD) is the most common dementia, characterized by significant cognitive impairments and neural network dysfunction. Currently, multiple therapeutic strategies are being developed to design effective anti-AD drugs. Among them, Withaferin A (WA), a natural steroidal lactone extracted from Withania somnifera leaves, has been shown to reduce amyloid-β (Aβ) peptide levels in vitro. However, its potential to improve cognitive function in AD remains unclear. Methods: In this study, 5xFAD mice were administered WA (2 mg/kg intraperitoneally every 2 days) for 14 days, and its neuroprotective effects were evaluated through behavioral tests, wide-field imaging, immunohistochemistry, and ELISA. Results: WA significantly improved short-term memory, as evidenced by enhanced performance in the Novel Object Recognition Test (NORT) (p < 0.001, n = 10), Novel Location Recognition Test (NLRT) (p < 0.01, n = 14), and Three-Chamber Social Test (TCST) (p < 0.001, n = 8). WA also ameliorated long-term memory deficits in the Morris Water Maze Test (MWMT) (p < 0.05, n = 7). Furthermore, cortical wide-field Ca2+ imaging revealed that WA treatment rescued slow-wave impairments by enhancing long-range coherence (0.8363 ± 0.0185, p < 0.01, n = 8) and reducing the frequency of slow-wave activity (0.6578 ± 0.0512 Hz, p < 0.01, n = 8). Additionally, WA treatment significantly reduced Aβ plaque deposition in both cortical and hippocampal regions. Conclusions: These findings suggest that WA may be a promising therapeutic agent for AD, exerting neuroprotective effects.
Keywords: Alzheimer’s; cognitive function; cortical slow-wave activity; neuroprotection; withaferin A.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures
Similar articles
-
Selegiline for Alzheimer's disease.Cochrane Database Syst Rev. 2003;(1):CD000442. doi: 10.1002/14651858.CD000442. Cochrane Database Syst Rev. 2003. PMID: 12535396
-
Co-Aggregation of Syndecan-3 with β-Amyloid Aggravates Neuroinflammation and Cognitive Impairment in 5×FAD Mice.Int J Mol Sci. 2025 Jun 8;26(12):5502. doi: 10.3390/ijms26125502. Int J Mol Sci. 2025. PMID: 40564963 Free PMC article.
-
Galantamine for Alzheimer's disease.Cochrane Database Syst Rev. 2002;(3):CD001747. doi: 10.1002/14651858.CD001747. Cochrane Database Syst Rev. 2002. Update in: Cochrane Database Syst Rev. 2004 Oct 18;(4):CD001747. doi: 10.1002/14651858.CD001747.pub2. PMID: 12137632 Updated.
-
Rationale for a Multi-Factorial Approach for the Reversal of Cognitive Decline in Alzheimer's Disease and MCI: A Review.Int J Mol Sci. 2023 Jan 14;24(2):1659. doi: 10.3390/ijms24021659. Int J Mol Sci. 2023. PMID: 36675177 Free PMC article.
-
Cholinesterase inhibitors for vascular dementia and other vascular cognitive impairments: a network meta-analysis.Cochrane Database Syst Rev. 2021 Feb 22;2(2):CD013306. doi: 10.1002/14651858.CD013306.pub2. Cochrane Database Syst Rev. 2021. PMID: 33704781 Free PMC article.
References
-
- Gueorguieva I., Willis B.A., Chua L., Chow K., Ernest C.S., Shcherbinin S., Ardayfio P., Mullins G.R., Sims J.R. Donanemab Population Pharmacokinetics, Amyloid Plaque Reduction, and Safety in Participants with Alzheimer’s Disease. Clin. Pharmacol. Ther. 2023;113:1258–1267. doi: 10.1002/cpt.2875. - DOI - PubMed
Grants and funding
LinkOut - more resources
Full Text Sources
Miscellaneous

