Synergistic Antitumor Effects of Caerin Peptides and Dendritic Cell Vaccines in a 4T-1 Murine Breast Cancer Model
- PMID: 40573908
- PMCID: PMC12197375
- DOI: 10.3390/vaccines13060577
Synergistic Antitumor Effects of Caerin Peptides and Dendritic Cell Vaccines in a 4T-1 Murine Breast Cancer Model
Abstract
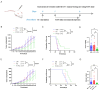
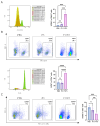
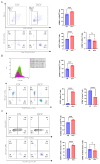
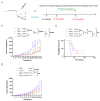
Background/Objectives: Breast cancer remains a leading cause of cancer-related mortality among women worldwide, necessitating novel therapeutic strategies. This study aimed to investigate the synergistic antitumor effects of caerin peptides (F1/F3) combined with dendritic cell (DC) vaccines in a 4T-1 murine breast cancer model, providing new insights for breast cancer immunotherapy. Methods: In vitro experiments evaluated the effects of F1/F3 on 4T-1 cell proliferation and apoptosis. A 4T-1 breast cancer mouse model was established, and treatments included F1/F3 alone, DC vaccines (DCV1: loaded with whole tumor antigens; DCV2: loaded with F1/F3-induced apoptotic antigens), or combination therapy. Flow cytometry analyzed immune cell subsets in the tumor microenvironment and lymph nodes, while ELISA measured cytokine levels. Results: F1/F3 significantly inhibited 4T-1 cell proliferation and induced apoptosis while suppressing tumor growth and lung metastasis in vivo. Flow cytometry revealed increased infiltration of CD4+ T cells and cDC1 in tumors, along with reduced PD-L1 expression. DCV2 exhibited stronger T-cell proliferation induction and lower IL-10 secretion in vitro. Combination therapy with DCV2 and F1/F3 demonstrated superior tumor suppression compared to monotherapy. Conclusions: F1/F3 enhances antitumor immunity by modulating the tumor microenvironment, and its combination with DCV2 yields synergistic effects. This study provides experimental evidence for combination immunotherapy in breast cancer, with potential for further optimization of DC vaccine design to improve efficacy.
Keywords: 4T-1; DC vaccine; DLNs; TME; caerin 1.1/1.9.
Conflict of interest statement
The authors J.L., Y.L. (Yuandong Luo) and Q.F. were employed by the Zhongao Biomedical Technology (Guangdong) Co., Ltd. The remaining authors declare that this research was conducted in the absence of any commercial or financial relationships that could be construed as potential conflicts of interest. The authors declare no conflict of interest.
Figures





References
-
- Onkar S.S., Carleton N.M., Lucas P.C., Bruno T.C., Lee A.V., Vignali D.A.A., Oesterreich S. The Great Immune Escape: Understanding the Divergent Immune Response in Breast Cancer Subtypes. Cancer Discov. 2023;13:23–40. doi: 10.1158/2159-8290.CD-22-0475. - DOI - PMC - PubMed
-
- Jacobs A., Lemoine A., Joshi G.P., Van de Velde M., Bonnet F., PROSPECT Working Group Collaborators PROSPECT guideline for oncological breast surgery: A systematic review and procedure-specific postoperative pain management recommendations. Anaesthesia. 2020;75:664–673. doi: 10.1111/anae.14964. - DOI - PMC - PubMed
LinkOut - more resources
Full Text Sources
Other Literature Sources
Research Materials
Miscellaneous

