Tissue Distribution and Pharmacokinetic Characteristics of Aztreonam Based on Multi-Species PBPK Model
- PMID: 40574060
- PMCID: PMC12196478
- DOI: 10.3390/pharmaceutics17060748
Tissue Distribution and Pharmacokinetic Characteristics of Aztreonam Based on Multi-Species PBPK Model
Abstract
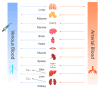
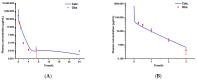
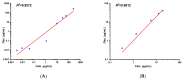
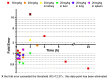
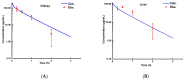
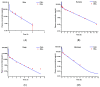
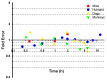
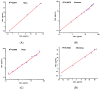
Background/Objectives: As a monocyclic β-lactam antibiotic, aztreonam has regained attention recently because combining it with β-lactamase inhibitors helps fight drug-resistant bacteria. This study aimed to systematically characterize the plasma and tissue concentration-time profiles of aztreonam in rats, mice, dogs, monkeys, and humans by developing a multi-species, physiologically based pharmacokinetic (PBPK) model. Methods: A rat PBPK model was optimized and validated using plasma concentration-time curves determined by liquid chromatography-tandem mass spectrometry (LC-MS/MS) following intravenous administration, with reliability confirmed through another dose experiment. The rat model characteristics, modeling experience, ADMET Predictor (11.0) software prediction results, and allometric scaling were used to extrapolate to mouse, human, dog, and monkey models. The tissue-to-plasma partition coefficients (Kp values) were predicted using GastroPlus (9.0) software, and the sensitivity analyses of key parameters were evaluated. Finally, the cross-species validation was performed using the average fold error (AFE) and absolute relative error (ARE). Results: The cross-species validation showed that the model predictions were highly consistent with the experimental data (AFE < 2, ARE < 30%), but the deviation of the volume of distribution (Vss) in dogs and monkeys suggested the need to supplement the species-specific parameters to optimize the prediction accuracy. The Kp values revealed a high distribution of aztreonam in the kidneys (Kp = 2.0-3.0), which was consistent with its clearance mechanism dominated by renal excretion. Conclusions: The PBPK model developed in this study can be used to predict aztreonam pharmacokinetics across species, elucidating its renal-targeted distribution and providing key theoretical support for the clinical dose optimization of aztreonam, the assessment of target tissue exposure in drug-resistant bacterial infections, and the development of combination therapy strategies.
Keywords: aztreonam; concentration-time profile; multi-species extrapolation; physiologically based pharmacokinetic model.
Conflict of interest statement
The authors declare no conflicts of interest.
Figures


Similar articles
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
Cost-effectiveness of using prognostic information to select women with breast cancer for adjuvant systemic therapy.Health Technol Assess. 2006 Sep;10(34):iii-iv, ix-xi, 1-204. doi: 10.3310/hta10340. Health Technol Assess. 2006. PMID: 16959170
-
Diagnostic test accuracy and cost-effectiveness of tests for codeletion of chromosomal arms 1p and 19q in people with glioma.Cochrane Database Syst Rev. 2022 Mar 2;3(3):CD013387. doi: 10.1002/14651858.CD013387.pub2. Cochrane Database Syst Rev. 2022. PMID: 35233774 Free PMC article.
-
Intravenous magnesium sulphate and sotalol for prevention of atrial fibrillation after coronary artery bypass surgery: a systematic review and economic evaluation.Health Technol Assess. 2008 Jun;12(28):iii-iv, ix-95. doi: 10.3310/hta12280. Health Technol Assess. 2008. PMID: 18547499
-
A rapid and systematic review of the clinical effectiveness and cost-effectiveness of paclitaxel, docetaxel, gemcitabine and vinorelbine in non-small-cell lung cancer.Health Technol Assess. 2001;5(32):1-195. doi: 10.3310/hta5320. Health Technol Assess. 2001. PMID: 12065068
References
Grants and funding
LinkOut - more resources
Full Text Sources
Research Materials

