Tunable methacrylated decellularized heart matrix: a versatile scaffold for cardiac tissue engineering
- PMID: 40574825
- PMCID: PMC12198208
- DOI: 10.3389/fbioe.2025.1579246
Tunable methacrylated decellularized heart matrix: a versatile scaffold for cardiac tissue engineering
Abstract
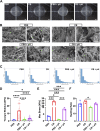
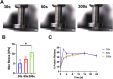
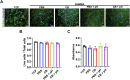
Therapeutic tissue regeneration remains a significant unmet need in heart failure and cardiovascular disease treatment, which are among the leading causes of death globally. Decellularized heart matrix (DHM) offer promising advantages for tissue engineering, including low immunogenicity and seamless integration into biological processes, facilitating biocompatibility. However, DHM is challenged by weak mechanical properties that limit its utility to biomedical applications like tissue engineering. To address this limitation, we functionalized DHM with methacryloyl functional groups (DHMMA) that support UV-induced crosslinking to enhance mechanical properties. By modulating the degree of methacryloyl substitution, a broad range of stiffness was achieved while maintaining cell viability on crosslinked DHMMA. Additionally, we show that increasing UV exposure time and pH increases DHMMA stiffness. Furthermore, topographical features transferred on DHMMA via soft lithography facilitated physical orientation of cells in culture. We demonstrate DHMMA as a scaffold with tunable stiffness and matrix-degradation properties suitable for cell survival and microfabrication for cardiac tissue engineering applications.
Keywords: biomaterials; decellularized heart matrix; matrix protein release; methacrylation; ultra-violet crosslinking.
Copyright © 2025 Pierre, Wu, Liu, Ertugral, Kothapalli and Senyo.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest. The author(s) declared that they were an editorial board member of Frontiers, at the time of submission. This had no impact on the peer review process and the final decision.
Figures




Similar articles
-
Nitrates for the prevention of cardiac morbidity and mortality in patients undergoing non-cardiac surgery.Cochrane Database Syst Rev. 2016 Aug 4;2016(8):CD010726. doi: 10.1002/14651858.CD010726.pub2. Cochrane Database Syst Rev. 2016. PMID: 27488764 Free PMC article.
-
Home treatment for mental health problems: a systematic review.Health Technol Assess. 2001;5(15):1-139. doi: 10.3310/hta5150. Health Technol Assess. 2001. PMID: 11532236
-
Signs and symptoms to determine if a patient presenting in primary care or hospital outpatient settings has COVID-19.Cochrane Database Syst Rev. 2022 May 20;5(5):CD013665. doi: 10.1002/14651858.CD013665.pub3. Cochrane Database Syst Rev. 2022. PMID: 35593186 Free PMC article.
-
Impact of residual disease as a prognostic factor for survival in women with advanced epithelial ovarian cancer after primary surgery.Cochrane Database Syst Rev. 2022 Sep 26;9(9):CD015048. doi: 10.1002/14651858.CD015048.pub2. Cochrane Database Syst Rev. 2022. PMID: 36161421 Free PMC article.
-
Immunogenicity and seroefficacy of pneumococcal conjugate vaccines: a systematic review and network meta-analysis.Health Technol Assess. 2024 Jul;28(34):1-109. doi: 10.3310/YWHA3079. Health Technol Assess. 2024. PMID: 39046101 Free PMC article.
References
LinkOut - more resources
Full Text Sources

