The efficacy and safety of chemoimmunotherapy in patients with MSI-L/MSS/pMMR status metastatic colorectal cancer: a systematic review and meta-analysis of randomized controlled trials
- PMID: 40575168
- PMCID: PMC12197930
- DOI: 10.3389/fonc.2025.1514485
The efficacy and safety of chemoimmunotherapy in patients with MSI-L/MSS/pMMR status metastatic colorectal cancer: a systematic review and meta-analysis of randomized controlled trials
Abstract
Background: Colorectal cancer (CRC) remains a leading cause of cancer-related mortality worldwide, necessitating innovative therapeutic approaches. Most patients with CRC exhibit microsatellite instability-low/stable (MSI-L/MSS) or proficient mismatch repair (pMMR) status, with chemotherapy being the standard first-line treatment. Chemoimmunotherapy, incorporating immune checkpoint inhibitors (ICIs), has emerged as a potential treatment for MSI-L/MSS/pMMR CRC. This study aimed to comprehensively evaluate the efficacy and safety of chemoimmunotherapy in metastatic CRC (mCRC) patients with MSI-L/MSS/pMMR status.
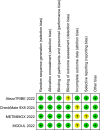
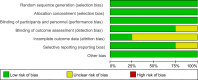
Methods: A systematic search of PubMed, EMBASE, ScienceDirect, and Cochrane Library was conducted in accordance with PRISMA guidelines, targeting studies published between May 2022 and September 2024. The meta-analyses utilized the generic inverse-variance method with a random effects model.
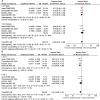
Results: Four studies encompassing 934 patients with mCRC met the inclusion criteria. The meta-analysis revealed a significant reduction in the risk of progression or death with chemoimmunotherapy compared with chemotherapy (HR: 0.82, 95% CI: 0.70-0.97, P = 0.02). Subgroup analyses based on sex (male vs. female) and ECOG status consistently demonstrated a significant benefit of chemoimmunotherapy in MSI-L/MSS/pMMR tumors. Adverse event analysis indicated an increase in adverse events in the chemoimmunotherapy group.
Conclusion: Existing evidence indicates a statistically significant and clinically meaningful benefit in PFS with chemoimmunotherapy, albeit with a slight increase in all-grade and high-grade toxicities compared to chemotherapy. Future research focusing on biomarkers and innovative treatments is essential for enhancing patient outcomes.
Systematic review registration: https://www.crd.york.ac.uk/PROSPERO/, identifier CRD42024520150.
Keywords: colorectal cancer; efficacy; immune checkpoint inhibitors; meta-analysis; safety.
Copyright © 2025 Zhang, Zhou, Hu, Pan, Luo and Yao.
Conflict of interest statement
The authors declare that the research was conducted in the absence of any commercial or financial relationships that could be construed as a potential conflict of interest.
Figures


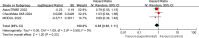

Similar articles
-
Evaluation of the efficacy and safety of first- and second-line immunotherapy in patients with metastatic colorectal cancer: a systematic review and network meta-analysis based on randomized controlled trials.Front Immunol. 2024 Sep 18;15:1439624. doi: 10.3389/fimmu.2024.1439624. eCollection 2024. Front Immunol. 2024. PMID: 39359729 Free PMC article.
-
The Efficacy and Safety of Durvalumab and Tremelimumab with Concomitant Treatment for MSS/pMMR Metastatic Colorectal Cancer: A Single Arm Meta-Analysis.J Gastrointest Cancer. 2025 Jan 29;56(1):56. doi: 10.1007/s12029-025-01181-4. J Gastrointest Cancer. 2025. PMID: 39875748
-
Certain pMMR colorectal cancer patients should undergo additional MSI-PCR testing to reduce the risk of misdiagnosing MSI-H and Lynch syndrome.BMC Cancer. 2025 Jul 1;25(1):1103. doi: 10.1186/s12885-025-14484-3. BMC Cancer. 2025. PMID: 40597865 Free PMC article.
-
Systemic pharmacological treatments for chronic plaque psoriasis: a network meta-analysis.Cochrane Database Syst Rev. 2021 Apr 19;4(4):CD011535. doi: 10.1002/14651858.CD011535.pub4. Cochrane Database Syst Rev. 2021. Update in: Cochrane Database Syst Rev. 2022 May 23;5:CD011535. doi: 10.1002/14651858.CD011535.pub5. PMID: 33871055 Free PMC article. Updated.
-
BRAF + EGFR +/- MEK inhibitors after immune checkpoint inhibitors in BRAF V600E mutated and deficient mismatch repair or microsatellite instability high metastatic colorectal cancer.Eur J Cancer. 2024 Oct;210:114290. doi: 10.1016/j.ejca.2024.114290. Epub 2024 Aug 22. Eur J Cancer. 2024. PMID: 39216175
References
Publication types
LinkOut - more resources
Full Text Sources
Miscellaneous

